
New Reaction Developed for Synthesis of Diazo Compounds Expands Pharmaceutical Development
New Synthetic Pathway for Diazo Compounds
A team of researchers from the Tokyo University of Science has made a significant breakthrough in synthetic chemistry, developing a novel reaction for the efficient synthesis of diazo esters from azido compounds. This new methodology not only enhances the safety of producing vital pharmaceutical intermediates but also opens new avenues in drug development involving nitrogen-containing heterocycles.
Importance of Diazo Compounds
Diazo esters are crucial building blocks in the synthesis of pharmaceuticals and functional molecules. However, traditional methods relied on hazardous reactants like diazomethane, which posed significant safety risks in laboratory settings. The recent innovation aims to provide a more practical and safer alternative, addressing the crucial need for safer synthetic techniques in organic chemistry.
Development of the Reaction
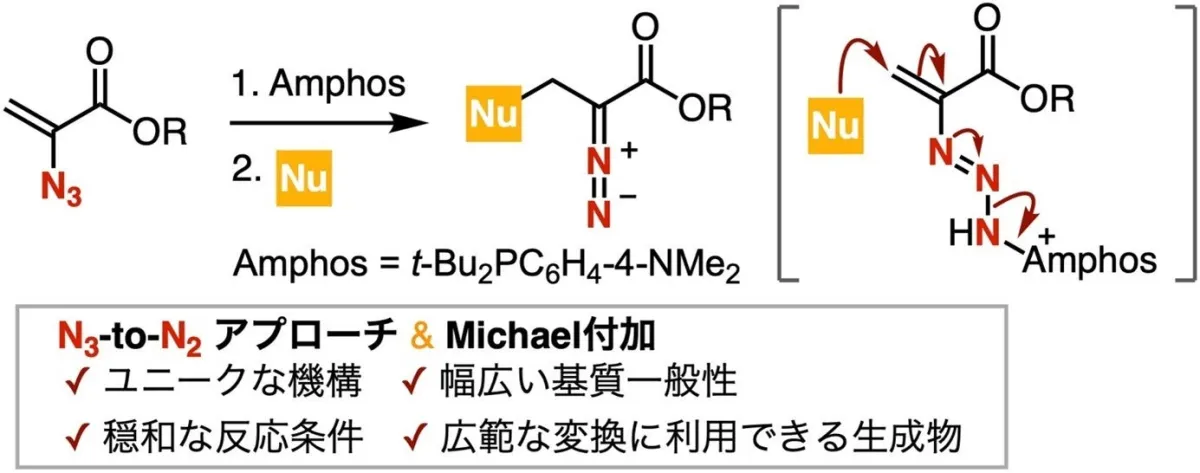
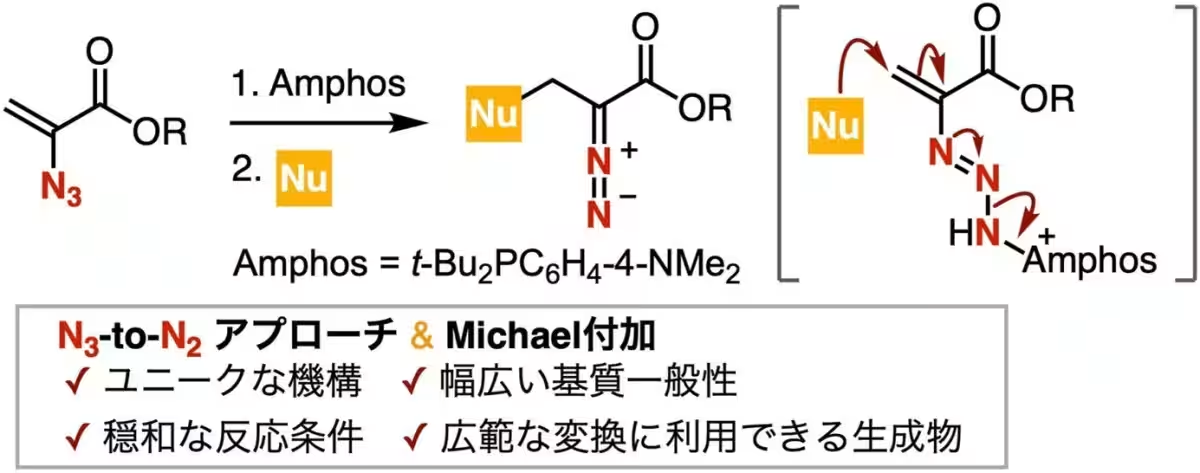
Led by Professor Suguru Yoshida, the research team has focused on utilizing 2-azidoacrylic acid esters coupled with phosphines to create a stable phosphazide intermediate. This intermediate facilitates a unique reaction sequence involving Michael addition and the cleavage of N-N bonds under mild conditions, leading to the direct synthesis of valuable diazo esters.
“Our goal was to exploit the stable nature of phosphazide intermediates in order to perform these reactions safely and efficiently. This technique not only simplifies the synthesis but also allows for a broader range of nucleophiles, such as thiols and amines, to be used,” said Yoshida.
The team has demonstrated that various diazo esters can be synthesized quickly and safely, establishing a foundation for future applications in the development of medicinal compounds.
A Versatile Approach
The method is adaptable and can be applied to a wide range of substrates. The team confirmed the successful synthesis of diazo compounds not only from primary and secondary thiols but also from cyclic and acyclic secondary amines. Notably, the reaction allows for substrates with diverse functional groups like bromo, hydroxyl, and ester groups, expanding its utility in organic synthesis.
In one case study, the team achieved the production of fluoxetine derivatives, illustrating the method’s practical potential. The reactions took place on a 10 mmol scale, showcasing the scalability of this innovative process.
Despite its many advantages, the reaction encountered limitations with substrates containing free carboxylic acids, indicating that careful selection of functional groups is essential for optimal results.
Detailed Mechanism
To understand the reaction mechanism thoroughly, the researchers isolated phosphine-imine by-products and conducted control experiments with TEMPO, alongside NMR measurements. These investigations revealed that the formation of the phosphazide intermediate is a driving force for both the Michael addition and the N-N bond cleavage.
In summary, the high basicity of the phosphazide intermediate is thought to be the critical factor contributing to its reactivity. The research outlines how this approach can yield diazo esters capable of further transformation through oxidation, reduction, rhodium catalyzed reactions, cycloaddition, and more, proving its applicability in creating nitrogen-containing heterocycles such as enamines, indoles, and pyrazoles.
Conclusion
Professor Yoshida expressed optimism regarding the impact of this discovery: "By maximizing the insights and techniques we've gained through our prior research, we've successfully uncovered a new method for utilizing azido groups. Given the widespread importance of diazo compounds in the synthesis of pharmaceuticals and functional molecules, we believe this method can contribute to a more practical and versatile synthesis of a wide variety of diazo compounds across diverse research areas."
This research was supported by Japan Society for the Promotion of Science Grants-in-Aid for Scientific Research (KAKENHI) and Asahi Glass Foundation.
For a detailed exploration of this research, the scholarly work has been published in the journal Angewandte Chemie International Edition.
Related Terms
1. Diazo Esters: Highly reactive compounds utilized extensively in pharmaceutical synthesis.
2. Azido Compounds: Chemical compounds containing the azide functional group, leading to valuable intermediates.
3. Phosphines: Organic compounds containing phosphorus, vital for creating phosphazide intermediates.
Topics Consumer Products & Retail)









【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.