

Discovering 4s Electrons in Oxygen Deficient TiO and VO: A New Mechanism of Metalization
Discovering 4s Electrons in Oxygen Deficient Titanium and Vanadium Oxides
Recent research conducted by a team from Waseda University and various institutions has led to a groundbreaking discovery in the realm of transition metal oxides. The findings reveal the presence of 4s electrons surrounding oxygen vacancies in rock salt-type titanium oxide (TiO) and vanadium oxide (VO). This research not only highlights a novel mechanism for metalization in materials previously classified as Mott insulators but also opens avenues for future applications in catalysis and materials science.
Key Findings
- - Existence of 4s Electrons: The study identified 4s electrons around oxygen vacancies in TiO and VO, challenging previous theories that suggested such electrons did not exist in transition metal oxides.
- - New Mechanism for Metalization: The research clarifies how a portion of the 3d electrons from the Mott-type TiO and VO transition to the 4s orbital, resulting in a metallic state rather than the expected insulating state.
- - Significance in Material Science: This discovery provides a new degree of freedom in controlling the physical and chemical properties of transition metal oxides, which could be significant for developing catalysts essential for a decarbonized society.
Background of the Study
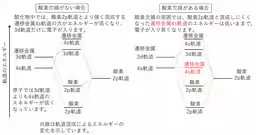
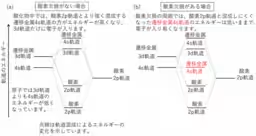
Historically, it's accepted that within atoms, 4s orbital electrons are generally of lower energy than 3d electrons. Elements like potassium and calcium demonstrate this with their electron configuration, filling the 4s orbital before the 3d. However, in transition metal oxides, where oxygen ions coordinate to transition elements, the energy levels change, and the 4s orbital typically experiences a higher energy state, making it less likely for these electrons to play a role in the material's properties.
Traditionally, transition metal oxides have been characterized by their 3d electrons, which dictate their magnetic and chemical behavior. However, the new findings reveal that 4s orbitals could also be playing a critical role in these materials.
Research Methodology
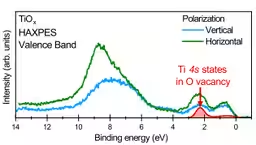
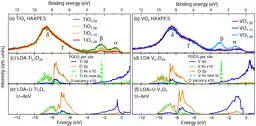
The research team utilized hard X-ray photoelectron spectroscopy (HAXPES) at the SPring-8 synchrotron facility to conduct detailed electronic structure measurements of TiO and VO. This technique allowed them to probe deeper into the properties of these materials, which are conventionally difficult to analyze due to the complexity of their electronic states.
Through their measurements, they observed significant peaks corresponding to the binding energies of 3d and 4s electrons. The results demonstrated that, contrary to previous assumptions, 4s orbitals are indeed present and can accommodate electrons in the oxygen-deficient rock-salt structure in transition metal oxides.
Implications of the Findings
The discovery emphasizes the potential to manipulate material properties by considering 4s electrons in transition metal oxides. Catalysts designed with these materials could be engineered to enhance efficiency in various chemical reactions, which is crucial for advancements in energy technologies and efforts towards sustainability.
Additionally, the research opens up discussions regarding the existence of 4s electrons in other transition metal oxides. Understanding whether this phenomenon occurs universally could lead to further revolutionary changes in how we design and utilize materials in technology.
Future Research Directions
While the findings are promising, the researchers point out that exploring the existence and roles of 4s electrons in a broader range of transition metal oxides remains a primary focus. This could drive innovations in materials that possess unique properties, potentially leading to the creation of novel conductive or magnetic materials.
The ongoing collaborative efforts between researchers in physics, chemistry, and materials science aim to unravel the complexities of electronic states in these compounds, potentially bridging the gap between various scientific fields.
Conclusion
The discovery of 4s electrons in oxygen-deficient TiO and VO signifies a paradigm shift in our understanding of transition metal oxides. By unveiling mechanisms that govern metalization in Mott insulators, this research not only contributes to academic knowledge but also has potential practical implications in advancing catalysis for a sustainable future. As researchers delve deeper into the realm of transition metal chemistry, the possibilities for innovative applications remain limitless.

Topics Consumer Technology)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.