

Mune Pharmaceutical Strengthens GMP Compliance Through i-Reporter Implementation
when, where, and by whom records were made, crucial for GMP compliance.
In 2024, Mune Pharmaceutical adopted i-Reporter, fostering a cross-departmental collaboration between the Quality Assurance and Manufacturing teams. This initiative included a one-month trial period to refine the documentation process, ultimately allowing for the digitization of essential records.
1. Enhanced GMP Compliance: The transition to i-Reporter enabled automatic recording of change history, timestamps, and account information, which diminished the risks of manipulation and ensured audit trails, thus reinforcing compliance with GMP.
2. Reduction in Data Entry Errors: The introduction of Bluetooth connectivity with measurement instruments allowed for automatic data transfers, significantly decreasing the manual entry errors from 20-30 instances annually to nearly zero.
3. Improved Document Management Efficiency: By employing automated calculations, the time needed for document inputs was halved, significantly mitigating the workload associated with record-keeping.
4. Streamlined Final Checks: The verification process time plummeted from 20 minutes to just 5 minutes, reducing the final confirmation duration by 75%, thus enhancing productivity.
5. Cost Savings on Paper Management: The digital transformation halved the storage space needed for records and decreased transportation costs related to document handling by about 70%, resulting in financial savings of approximately 320,000 yen annually.
Looking ahead, Mune Pharmaceutical plans to utilize i-Repo Link and IoT DataShare to further automate their recording processes by integrating PLC systems, aiming for comprehensive enhancements in GMP compliance and operational efficiency. Automatic reflection of process data, such as temperature and pressure on documents, is expected to facilitate even greater improvements in field operations.
To learn more about Mune Pharmaceutical's successful i-Reporter implementation, visit the following link: i-Reporter Case Study
CIMTOPS’ system, i-Reporter, is utilized by over 4,500 companies and 220,000 users across Japan, establishing itself as the leading solution for field report digitization. With features that allow for easy integration with existing paper and Excel formats, i-Reporter continues to revolutionize documentation processes across various industries, contributing to significant labor and time efficiencies.


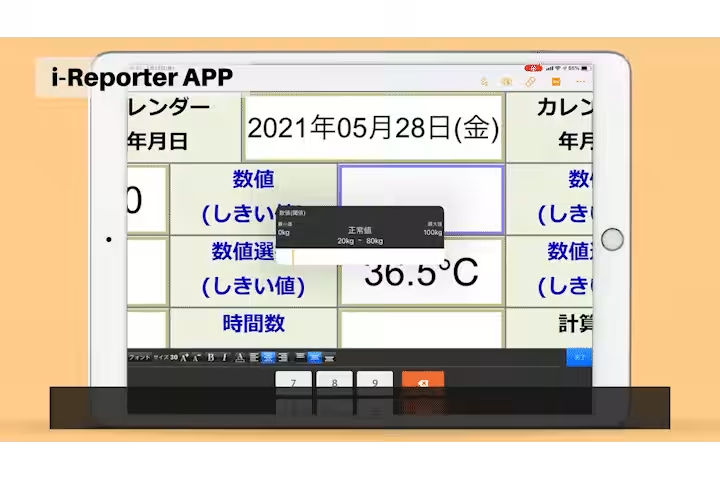
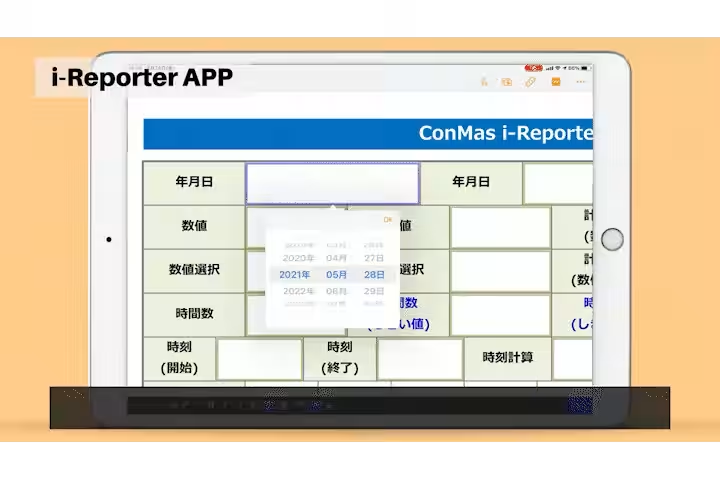
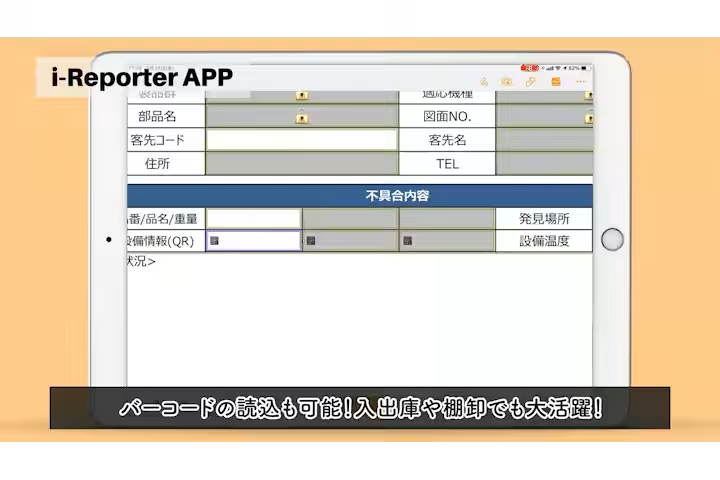
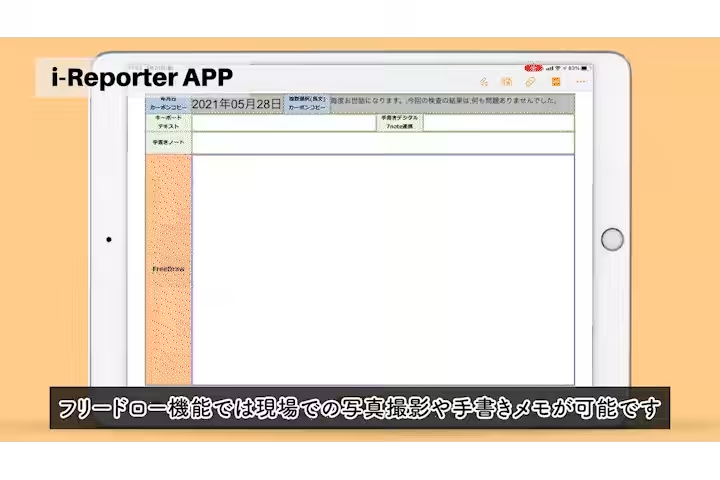







- - Manual Data Entry Errors: Frequent manual data entry errors, particularly in the repetition of weight measurements, led to 20-30 mistakes annually.
- - High Costs Associated with Paper Management: The need for transporting, collecting, and storing physical documents incurred around 430,000 yen annually in costs related to labor and material.
- - Inefficient Final Checks: The final verification process required about 20 minutes, as staff had to review several paper documents manually.
In 2024, Mune Pharmaceutical adopted i-Reporter, fostering a cross-departmental collaboration between the Quality Assurance and Manufacturing teams. This initiative included a one-month trial period to refine the documentation process, ultimately allowing for the digitization of essential records.
Implementation Highlights
- - i-Reporter Adoption: The digital transformation was seamless, maintaining existing documentation while fulfilling the necessary CSV requirements for GMP.
- - Cross-Departmental Collaboration: Coordination between departments ensured that GMP requirements were met operationally, improving efficiency and compliance.
- - Phased Deployment: Initial trials of the system allowed for gradual expansion based on feedback from the field, which proved crucial for user adoption and system enhancement.
Key Benefits and Effects
1. Enhanced GMP Compliance: The transition to i-Reporter enabled automatic recording of change history, timestamps, and account information, which diminished the risks of manipulation and ensured audit trails, thus reinforcing compliance with GMP.
2. Reduction in Data Entry Errors: The introduction of Bluetooth connectivity with measurement instruments allowed for automatic data transfers, significantly decreasing the manual entry errors from 20-30 instances annually to nearly zero.
3. Improved Document Management Efficiency: By employing automated calculations, the time needed for document inputs was halved, significantly mitigating the workload associated with record-keeping.
4. Streamlined Final Checks: The verification process time plummeted from 20 minutes to just 5 minutes, reducing the final confirmation duration by 75%, thus enhancing productivity.
5. Cost Savings on Paper Management: The digital transformation halved the storage space needed for records and decreased transportation costs related to document handling by about 70%, resulting in financial savings of approximately 320,000 yen annually.
Future Prospects
Looking ahead, Mune Pharmaceutical plans to utilize i-Repo Link and IoT DataShare to further automate their recording processes by integrating PLC systems, aiming for comprehensive enhancements in GMP compliance and operational efficiency. Automatic reflection of process data, such as temperature and pressure on documents, is expected to facilitate even greater improvements in field operations.
To learn more about Mune Pharmaceutical's successful i-Reporter implementation, visit the following link: i-Reporter Case Study
Company Overview
Mune Pharmaceutical Co., Ltd.
- - Founded: 1909
- - Headquarters: 859 Ozaki, Awaji City, Hyogo Prefecture, 656-1501 Japan
- - Business: Pharmaceutical manufacturing and sales
- - Corporate Philosophy: Sincerity and Compassion
- - Official Website: Mune Pharmaceutical
About CIMTOPS, Inc.
- - Company Name: CIMTOPS, Inc.
- - Headquarters: 2-2, Kamioozaki, Shinagawa, Tokyo, 141-0021, Japan
- - CEO: Takashi Mizuno
- - Established: October 1, 1991
- - Capital: 165 million yen
- - Sales: 2.087 billion yen (FY2024)
- - Employees: 88 (as of September 2025)
- - Company Website: CIMTOPS
CIMTOPS’ system, i-Reporter, is utilized by over 4,500 companies and 220,000 users across Japan, establishing itself as the leading solution for field report digitization. With features that allow for easy integration with existing paper and Excel formats, i-Reporter continues to revolutionize documentation processes across various industries, contributing to significant labor and time efficiencies.













Topics Consumer Technology)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.