
Ceglu™ Receives FDA Drug Master File Registration for Stem Cell Research
Ceglu™ Secures FDA Drug Master File Registration
Sekisui Chemical Co., led by President Ikusuke Shimizu, has proudly announced the completion of registration of its innovative cell adhesion polymer, Ceglu™, with the U.S. Food and Drug Administration (FDA). This milestone allows Ceglu™ to be recognized as a suitable raw material for applications in clinical trials and the manufacturing of regenerative medicine products, including cell and gene therapies in the United States. This registration is expected to enhance the development and commercialization of regenerative medicine.
Significance of the DMF Registration
The Drug Master File (DMF) system serves as a comprehensive registry of detailed information related to the components and manufacturing processes of drugs and regenerative medicine products. With the DMF registration, pharmaceutical companies and biotech firms utilizing Ceglu™ can reference this master file in their IND (Investigational New Drug) and BLA (Biologics License Application) submissions. The anticipated benefits of this registration include:
- - Reduced Reporting Burden to Regulatory Authorities: This allows companies to streamline their compliance processes and focus on innovation.
- - Increased Trust in Quality and Safety Information: The registration offers a reliable source of information, enhancing confidence among stakeholders.
- - Facilitated and Accelerated Development and Approval Processes: This assurance is pivotal for companies working fast-paced environments to bring innovative therapies to market.
Ceglu™ is expected to promote wider applications from research involving stem cell cultures, such as iPS cells, to clinical and commercial manufacturing phases. The registration signifies Ceglu™’s potential to offer safety, reproducibility, and sustainability in various research applications, contributing significantly to the acceleration of practical regenerative medicine solutions in the world’s largest drug market, the United States.
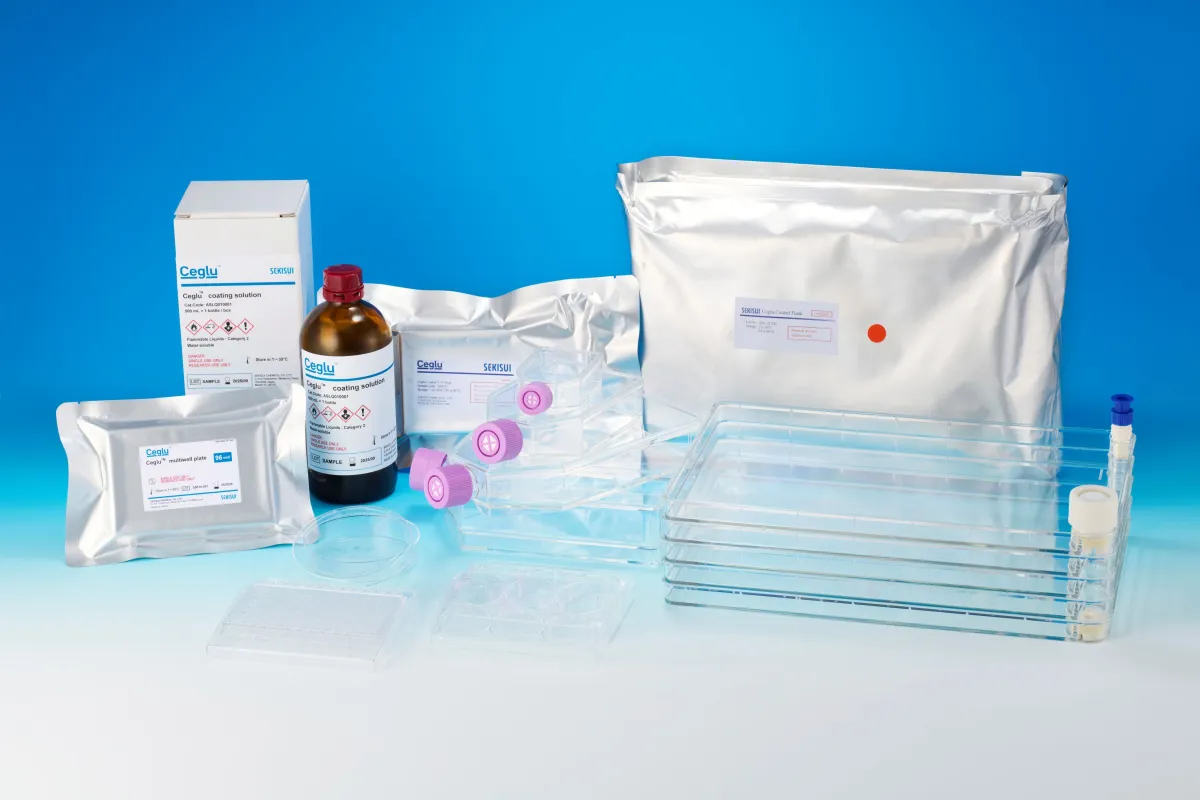
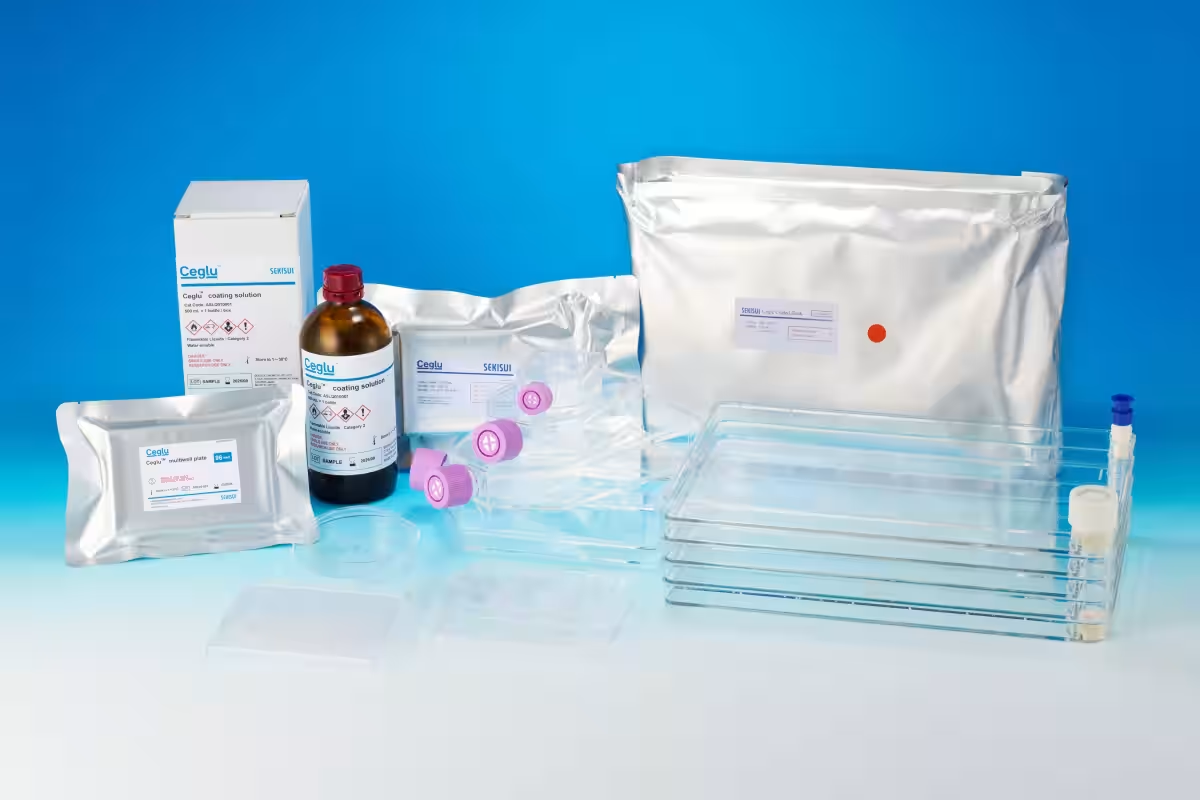
Ceglu™ Product Lineup
Ceglu™ is a chemically synthesized polymer that enables the culture of iPS cells and can coat various material surfaces when turned into a solution. It boasts higher stability at room temperature compared to proteins, eliminating the need for cold-chain logistics—essential for maintaining quality in sensitive applications. Additionally, it contains no animal-derived components, ensuring high safety standards. Ceglu™ has successfully passed safety tests compliant with both the United States Pharmacopeia and the Japanese Pharmacopeia.
In pursuit of a comprehensive cell adhesion platform from lab-scale to manufacturing-scale, Sekisui Chemical offers Ceglu™ coating solutions alongside Ceglu™ cultureware—plates that have been pre-coated. Notably, Ceglu™ has been harnessed in numerous applications, including enhancing the reproducibility of cardiac differentiation from iPS-derived cardiomyocytes, and optimizing cell culture quality with automated systems. The polymer can also enable scaling up cell cultures via coating on nonwoven fabrics and microcarriers.
For more information, visit the Ceglu™ product page: Ceglu™ Product Page.
Conclusion
Ceglu™'s recent FDA registration places it at the forefront of advancements in regenerative medicine, addressing the growing need for reliable materials that support innovation in cellular therapies. As Sekisui Chemical advances this promising platform, Ceglu™ is poised to play an integral role in shaping the future landscape of regenerative treatments and techniques worldwide.
Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.