
Rosetta Unveils Cutting-Edge AI, Reducing CSR Draft Time by Over 90%
Revolutionizing Clinical Study Reports with AI
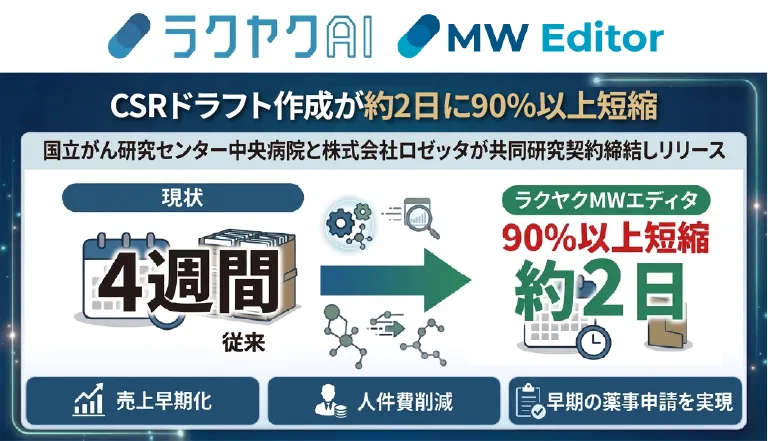
In the fast-paced world of pharmaceuticals, every moment counts, especially in the development of new drugs. A crucial component of this process is the Clinical Study Report (CSR), which outlines every detail of clinical trials. Traditionally, crafting these essential documents has been a time-consuming endeavor, often taking around four weeks just to draft. However, this is all changing thanks to a breakthrough development from Rosetta, a major player in AI translation services in Japan.
The Challenge of Traditional CSR Development
The CSR is a highly specialized document that must adhere to the strict guidelines set by ICH E3. Typically, a single drug development project may require 10 to 20 CSRs, each sometimes exceeding 100 pages. The lengthy process involves multiple stages, including:
- - Data organization
- - Narrative writing
- - Quality checks (QC)
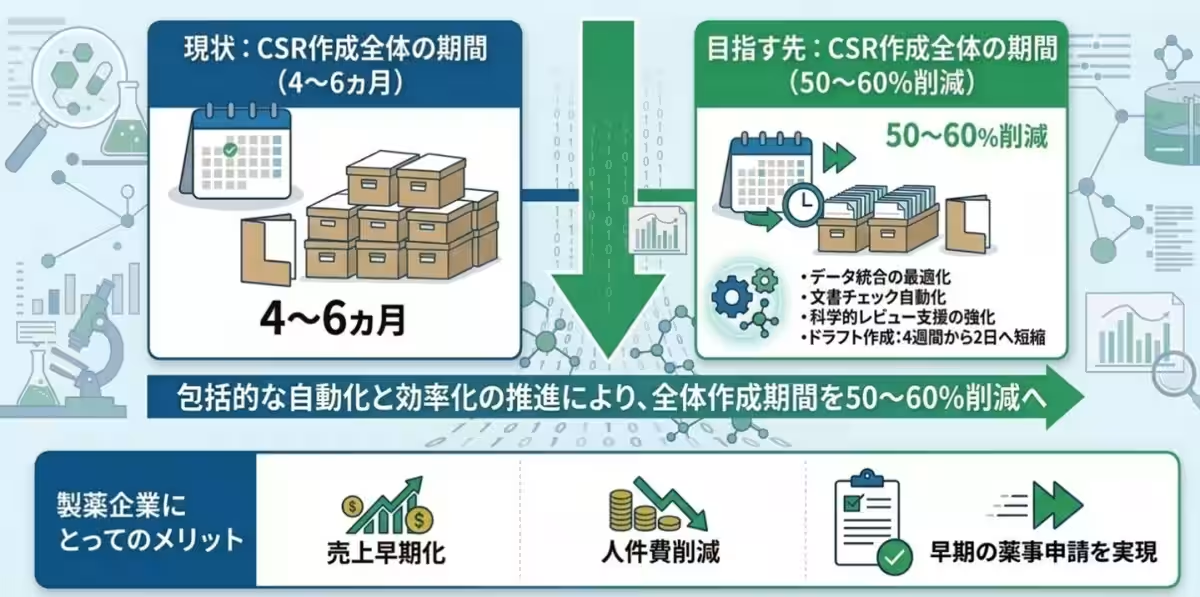
As a result, completing a CSR can take anywhere from four to six months, placing a significant burden on pharmaceutical companies. Recognizing the need for a more efficient solution, Rosetta has introduced an AI-driven tool designed to significantly cut down the drafting time.
Introducing the Rakuyaku MW Editor
Rosetta's latest innovation, the Rakuyaku MW Editor, is a cutting-edge writing support tool specifically developed for CSR, clinical trial protocols, and related documents. With an increasing demand for speed and quality in drug development, this tool harnesses the power of AI to assist medical writers, aiming to reduce both costs and time.
Key Features of the Rakuyaku MW Editor:
- - Automatic Draft Creation: Users can upload source documents, and the tool generates draft content chapter by chapter.
- - Reference Tracking: The generated text can show which parts of the source documents were used, aiding clarity and verifiability.
- - Alternative Suggestions: If desired, the AI provides different versions of generated text for comparison.
- - Editing Tools: The editor allows modifications including font design, size, and layout.
- - AI Chatbot: For additional support, an integrated chatbot is available.
This innovative editor significantly enhances efficiency in the drafting process while ensuring uniform quality across documents. Following validation by experienced in-house medical writers, the tool has successfully reduced the draft period from approximately four weeks to merely two days, achieving over 90% labor savings for companies that have begun utilizing it.
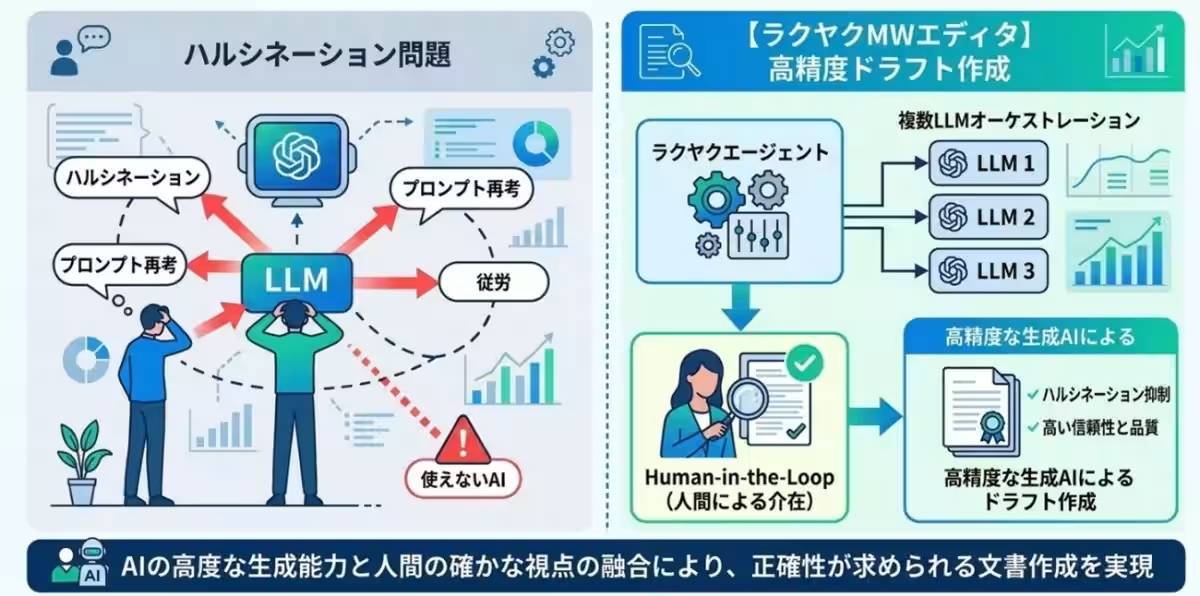
Addressing Concerns About AI Accuracy
One common challenge with AI is the concern over �201challenges associated with inaccuracies, often termed �201challucinations. However, Rosetta has implemented solutions to combat these issues through:
- - Multiple LLM Integration: Leveraging various language model engines to ensure accuracy.
- - Human-in-the-Loop Process: Involving human oversight throughout the drafting process guarantees high-quality and reliable results.
This hybrid design allows the tool to maintain stringent quality standards required in pharmaceutical documentation.
Future Prospects: Transforming CSR Creation
Looking forward, Rosetta aims not just to enhance the CSR drafting speed but to transform the entire CSR creation process, targeting a reduction in total preparation time by 50-60%. This innovation could lead to earlier drug submissions and expedite market availability, reshaping the landscape of pharmaceutical document preparation and significantly contributing to advancements in healthcare.
About Rakuyaku AI
Rosetta's Rakuyaku AI platform is specifically tailored for the pharmaceutical sector, providing solutions for automatic CSR generation, document consistency checks, and translations, all designed to streamline operations and reduce costs for pharmaceutical companies. By significantly shortening the complex documentation processes, Rakuyaku AI plays a vital role in accelerating the introduction of both generic and brand-name drugs to the market.
For more details about Rakuyaku AI, you can visit Rakuyaku AI Product Site, download informative resources at Rakuyaku AI Downloads, or make inquiries at Rakuyaku AI Inquiries.
About Metarial Group
The Metarial Group is committed to liberating individuals from constraints of place, time, and language, positioning itself as a leader in the translation market. They offer customized AI development services across various sectors, including legal, pharmaceutical, financial, and IT industries, supporting a wide range of over 2,000 fields. For more information, visit Metarial Group.
Conclusion
Rosetta's innovative use of AI demonstrates the significant potential for technology to streamline key processes in the pharmaceutical industry, paving the way for faster drug development and ultimately improving patient access to essential medications.

Topics Business Technology)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.