
Smith & Nephew Launches Innovative CATALYSTEM Hip Implant in Japan
Smith & Nephew Introduces CATALYSTEM in Japan
Smith & Nephew, a global leader in medical technology, has officially launched the CATALYSTEM femoral implant in Japan. This state-of-the-art device aims to address significant challenges faced during hip replacement surgeries, including the risk of perioperative fractures associated with traditional implants.
Background on the Challenges of Hip Replacement Surgeries
Over recent years, the number of hip replacement surgeries in Japan has steadily risen, and the use of cementless femoral implants has become increasingly common. However, one of the notable complications in this procedure is the occurrence of bone fractures around the implant, either during or after surgery. Such incidents may necessitate additional surgeries for implant revision or fracture fixation, presenting a substantial challenge for patients and surgeons alike. Reports indicate that bone fractures are the most frequent complication linked to hip surgeries, underscore the urgency of addressing this issue.
The Solution: CATALYSTEM’s Design Features
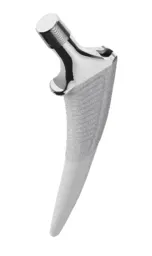
The CATALYSTEM implant is designed with a unique triple-taper stem design. This innovative approach minimizes distal dimensions while enhancing uniform load transfer to the proximal area, increasing the intramedullary fill. In consideration of the diverse morphology of patients around the world, including shorter stem lengths suitable for Japanese patients, the CATALYSTEM is available in a variety of sizes to accommodate different anatomies. This planning aligns with Smith & Nephew's commitment to providing specialized solutions tailored to local patient needs.
Furthermore, CATALYSTEM employs the proprietary ACCUBROACH™ design, which significantly reduces the risk of intraoperative fractures during insertion, compared to traditional instrumentation. High reproducibility between the broach and the implant has been demonstrated, achieving predictable stem placement and high rotational stability, which are critical for the longevity of hip arthroplasty.
Optimizing Patient Outcomes
The introduction of CATALYSTEM is anticipated to extend the endurance of hip implants, improving the quality of life for patients post-surgery. With its focus on minimizing surgical complications, Smith & Nephew aims to uphold its mission of enhancing patient care in Japan.
Smith & Nephew’s Commitment
Drawing from its experience and success around the globe, Smith & Nephew is dedicated to delivering products that best fit the needs of Japanese healthcare professionals and patients. With the launch of CATALYSTEM, the company aims to contribute positively to the evolution of hip replacement surgery.
Product Information
- - Name: CATALYSTEM Hip System
- - Approval Number: 30700BZX00112000
Company Insights
Smith & Nephew, established in Hull, UK in 1856, operates in over 100 countries and has 18,000 employees dedicated to advancing the field of orthopedics and wound care. The company's future revenue projection for 2025 stands at $5.8 billion. It is also listed on the FTSE 100.
For further information, please visit:

Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.