
Expansion of Production Facilities for Steering Microcatheters by Sumitomo Bakelite
Expansion of Production Facilities for Steering Microcatheters
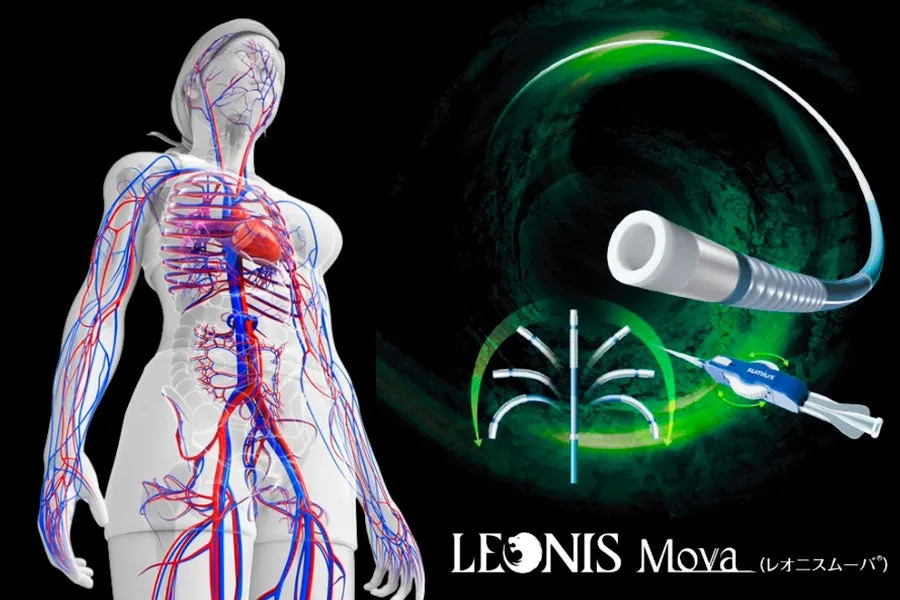
Sumitomo Bakelite Co., Ltd. has recently made an important decision to expand its production facilities located at Akita Sumitomo Bakelite Co., Ltd., in response to the increasing demand for steering microcatheters in vascular treatments. This innovative device allows for precise control of its tip direction from the operator’s hand, significantly reducing procedure time. Since its launch in 2016, the steering microcatheter has accumulated clinical records in various healthcare settings, both domestically and internationally, as the complexity and minimally invasive aspects of vascular treatments have advanced.
Changing Landscape of Vascular Treatments
As the aging population grows, the risks associated with vascular diseases such as atherosclerosis and vessel stenosis are rising annually. Additionally, there is an increasing demand for minimally invasive treatments, including vascular interventions, as viable options for elderly patients and those with comorbidities. These low-invasive procedures are gaining traction due to their reduced physical burden compared to surgical operations, leading to shorter hospitalization periods and improved quality of life. Among the essential devices for such treatments are microcatheters, which are utilized across a wide range of areas, including neurovascular, abdominal, and peripheral vascular applications. With the evolution of surrounding devices, the performance requirements for microcatheters are also on the rise, demanding higher precision for increasingly complex cases.
Features and Clinical Value of Steering Microcatheters
The steering microcatheter is designed with a unique structure that enables the operator to control the tip direction at will. This capability allows for a selective and stable insertion into highly curved vessels and complex vascular branches, which were challenging with traditional microcatheters. It is being utilized in various fields, such as embolization for tumors like hepatocellular carcinoma in abdominal applications, coiling procedures for cerebral aneurysms in the neurovascular domain, and treatment for ischemic diseases in peripheral vascular areas.
The movable tip functionality enhances accessibility to target vessels, facilitating easier selection while reducing the operators' workload and contributing to shorter procedure times. This results in decreased fluoroscopy times and a reduction in the number of imaging procedures required, ultimately leading to lower exposure for both patients and medical staff.
The steering microcatheter (Approval Number: 22600BZX00482000) known as Leonis Mover® has seen its indications expand significantly since its market entry. To align with changes in clinical needs and advancements in medical technology, the company continuously works on expanding the size varieties and improvements of its products. Domestically, approval for intracranial vascular treatment was achieved in 2022, and the insurance coverage has been broadened, reflecting the product's alignment with clinical demands. These characteristics have established the steering microcatheter as a reliable medical tool for safer and more assured vascular treatments, continuing to see a steady increase in demand, even a decade after its launch.
Overview of the Production Facility Expansion
The recent expansion of production facilities aims to ensure stable supply to healthcare settings while addressing the growing demand both locally and internationally. The preparation for integrating the new equipment will commence in April 2026, with operations expected to start in February 2027.
Domestically, the steering microcatheter is marketed by SB Kawasumi Corporation, which operates under the Sumitomo Bakelite group. Internationally, it is sold through partner companies across approximately 40 countries, with growing demand primarily from the United States and China. With the enhancement of production capacity, an increase to 200% in production capabilities is expected by the 2027 fiscal year compared to 2025, paving the way for expansion into new markets.
Looking Ahead
The Sumitomo Bakelite group is committed to responding to the evolving medical needs associated with minimally invasive treatments, working to resolve the challenges faced by both patients and healthcare professionals. The expansion of production facilities is part of their responsibility as a supplier of medical devices used in low-invasive treatments, with ongoing efforts to ensure stable supply and quality improvement, contributing to the sustainable development of healthcare.
For Product Inquiries
SB Kawasumi Corporation, Domestic Sales Division, Endovascular Product Sales Team
TEL: 03-5462-4824
SB Kawasumi Website
Contact Form


Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.