
Cambrex Accelerates Its Expansion in the US and Europe with New Facilities
Cambrex's Strategic Expansion in the US and Europe
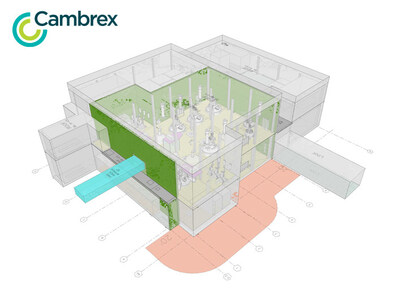
Cambrex, a prominent global Contract Development and Manufacturing Organization (CDMO), is taking significant strides in expanding its reach in the United States and Europe. Recently, the company announced the successful completion of initial engineering studies for a new large-scale active pharmaceutical ingredient (API) manufacturing plant in Charles City, Iowa. This development is a pivotal part of their previously announced $120 million investment aimed at enhancing their manufacturing capabilities in the US. The groundbreaking for this new facility is anticipated to take place by the end of 2026.
The Charles City expansion will feature a state-of-the-art plant with a capacity of 140,000 liters. This will include high- and medium-scale reactors, advanced Hastelloy agitated dryers, and improvements to existing manufacturing infrastructure. Following completion of this expansion, the site will see a significant 20% increase in its large-scale production capacity. The facility is designed to handle complex chemistry, including controlled substances, highly potent active pharmaceutical ingredients (HPAPIs), and commercial-scale liquid peptide synthesis. Such advancements position Cambrex as a trusted partner for pharmaceutical innovators looking for reliable manufacturing solutions.
In addition to its US expansion, Cambrex is also making considerable investments in Europe. The company has embarked on a $30 million expansion at its facility in Milan, Italy. This project promises to introduce new analytical development and process research capabilities, while also modernizing various production plants. The expansion in Milan is expected to wrap up by the second half of 2027, with plans to acquire additional land to support future growth and meet the rising demand for CDMO services.
Claudio Russolo, the Operations Director at Cambrex, shared the significance of their Milan site, stating, “Our facility in Milan has a long-standing tradition of supporting pharmaceutical development and manufacturing, and this year marks its 80th anniversary. We are committed to continuing our investments to meet the increasing market demand for complex chemistries, including peptides, highly potent molecules, and controlled substances.”
Cambrex’s ongoing investments are a testament to its commitment to delivering modernized solutions for the development and manufacturing of complex small molecules. The company’s scientific approach and dedication to operational excellence ensure that Cambrex remains at the forefront of supporting the next generation of life-changing treatments.
About Cambrex
Cambrex is a leading global Contract Development and Manufacturing Organization (CDMO) that provides pharmaceutical ingredient development and manufacturing services throughout the drug lifecycle, alongside comprehensive analytical and supporting services for new drug research. With over 45 years of experience and a team of more than 2,000 experts, Cambrex serves international clients across North America and Europe. The company offers a wide array of specialized technologies and capabilities in pharmaceuticals, including continuous flow, controlled substances, peptide synthesis, solid-state science, material characterization, and highly potent APIs.

Topics Consumer Products & Retail)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.