
Discovery of Negative Thermal Expansion Phenomena in Hydrogen Storage Materials
Discovery of Negative Thermal Expansion Phenomena in Hydrogen Storage Materials
Overview
In most materials around us, an increase in temperature generally leads to expansion, while a decrease leads to contraction. This characteristic is known as positive thermal expansion and is commonly observed in metals and plastics. However, some materials behave inversely, contracting with an increase in temperature. These materials are identified as negative thermal expansion (NTE) materials. The application of NTE materials can control thermal expansion coefficients when combined with positive thermal expansion materials. This has significant implications for modern industries, especially for electronic devices where precise thermal control is essential.
A research team, including Associate Professor Yoshikazu Mizuguchi from the Graduate School of Science at Tokyo Metropolitan University, graduate student Yuto Watanabe, Professor Takayoshi Katase from the Frontier Material Research Institute at Tokyo Science University, and Professor Akira Miura from Hokkaido University, has identified a unique phenomenon. They discovered that the hydrogenated transition metal zirconide CoZr2H3.49 exhibits negative thermal expansion exclusively along the c-axis in a specific temperature range below the Curie temperature (139 K).
The non-hydrogenated CoZr2 exhibits approximately a superconducting transition temperature of 6 K and displays negative thermal expansion along the c-axis over a wide temperature range. While it was known that CoZr2 transforms into a ferromagnet by absorbing hydrogen, the possibility of similar negative thermal expansion phenomena in CoZr2 has not been reported until this study. This research's main focus was on CoZr2H3.49, leading to the observation of its negative thermal expansion in the ferromagnetic region. The NTE exhibited by CoZr2H3.49 is driven by changes in the electronic state of the material, specifically by the magnetization transition below the Curie temperature.
Future studies may enable the maximization of the linear thermal expansion coefficient along the c-axis and provide control over the a-axis coefficient by varying the hydrogen content, paving the way for the development of zero thermal expansion materials using a single substance. This research was published in the Journal of the American Chemical Society on February 11 (Japan time) and is supported by various research funds, including JST's ERATO project and Tokyo Metropolitan University's young researcher support program.
Key Points
- - Negative Thermal Expansion Discovery: The NTE phenomenon was identified in CoZr2H3.49 along the c-axis.
- - Mechanism of NTE Variation: Unlike CoZr2's NTE, that of CoZr2H3.49 occurs below the Curie temperature and is driven by a ferromagnetic transition.
- - Distinct Mechanism: The mechanism driving the c-axis NTE in CoZr2H3.49 differs from that of the hydrogen-free CoZr2.
Background of the Research
In today's rapidly evolving industries, the control of thermal expansion coefficients is crucial to prevent deformation of materials and strain at interfaces between different kinds of materials. NTE materials are particularly significant because they can contract as temperature increases, offering a means to mitigate thermal expansions across various applications like semiconductor packaging and precision optical devices.
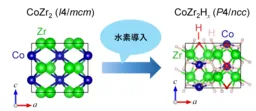
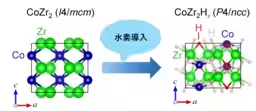
The discovery of c-axis negative thermal expansion by the research group led by Associate Professor Mizuguchi in 2022 has sparked further investigations, unveiling NTE in systems where Co has been replaced with another transition metal. The ability of transition metal zirconides to absorb hydrogen has further drawn attention towards their properties as hydrogen storage materials. Absorbing hydrogen can significantly alter a material's characteristics; specifically, the CoZr2 superconductor undergoes a major electronic state change upon hydrogen absorption, transitioning to a weak itinerant ferromagnetic state at low temperatures.
Additionally, the crystal structure largely remains unchanged during hydrogen absorption, shifting from the tetragonal I4/mcm (#140) space group to the P4/ncc (#130) space group. Given that the crystal structures of CoZr2H3.49 and CoZr2 exhibiting c-axis NTE are nearly identical, it was anticipated that negative thermal expansion would also manifest in CoZr2H3.49, emphasizing the importance of detailed study.
This research utilized synchrotron X-ray diffraction measurements to evaluate thermal expansion characteristics of CoZr2H3.49 within the temperature range below the Curie temperature.
Research Methodology
The study involved synthesizing CoZr2 powder and introducing hydrogen by heating it in a hydrogen atmosphere. The resulting hydrogenated powder was then sintered under high-pressure and high-temperature conditions to produce CoZr2H3.49 samples. The hydrogen amount was determined using temperature-programmed desorption gas analysis (TDS).
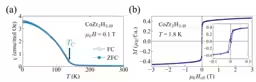
Magnetic susceptibility measurements of CoZr2H3.49 displayed a dramatic increase below the Curie temperature TC = 139 K, indicating a transition to ferromagnetic states. Magnetic behavior under applied fields reflected characteristics typical of weak itinerant ferromagnetic states.
Detailed Measurement Results
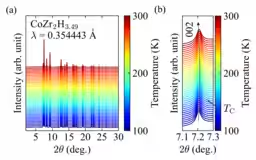
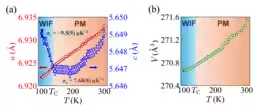
X-ray diffraction patterns were assessed across a temperature range from 300 K to 100 K. Although no structural changes were evident, the diffraction angles reflecting the lattice constant c exhibited a shift during cooling, suggesting a complex temperature-dependent behavior.
Reitveld analysis was used to extract temperature dependencies for the lattice constants a and c. Above the Curie temperature, the c-axis showed positive thermal expansion, but below this temperature, it demonstrated negative thermal expansion in weak itinerant ferromagnetic states.
Significance of the Research
This study successfully observed negative thermal expansion along the c-axis in CoZr2H3.49, classified as occurring below the Curie temperature in weak ferromagnetic phases. The distinct mechanism of NTE between CoZr2H3.49 and hydrogen-free CoZr2 suggests a unique phase transition type. Future research aimed at controlling hydrogen content may facilitate fine-tuning of thermal expansion coefficients and contribute to the development of zero thermal expansion materials from single compounds.
Topics General Business)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.