
Free Release of the China Pharmaceutical Innovation White Paper 2026 by SUGENA
Unveiling the China Pharmaceutical Innovation White Paper 2026
In a significant move for the pharmaceutical industry, SUGENA Co., Ltd. has released the China Pharmaceutical Innovation White Paper 2026 for free. This comprehensive report analyzes the current landscape of the life sciences ecosystem in China, highlighting the accelerating partnerships between global mega-pharma companies and Chinese biotech firms as they approach the impending patent cliff of 2030.
The Importance of Focusing on China’s Drug Discovery
The global pharmaceutical industry is at a pivotal point due to the expiration of patents on key products. As major pharmaceutical companies in Europe, America, and Japan scramble to replenish their pipelines, they are increasingly looking toward China for innovative assets. Over the past decade, China's biotech sector has witnessed remarkable growth in both quality and speed, making it a key player in the global market. This white paper organizes this market shift with objective data, making it an essential resource for industry stakeholders.
Key Insights from the White Paper
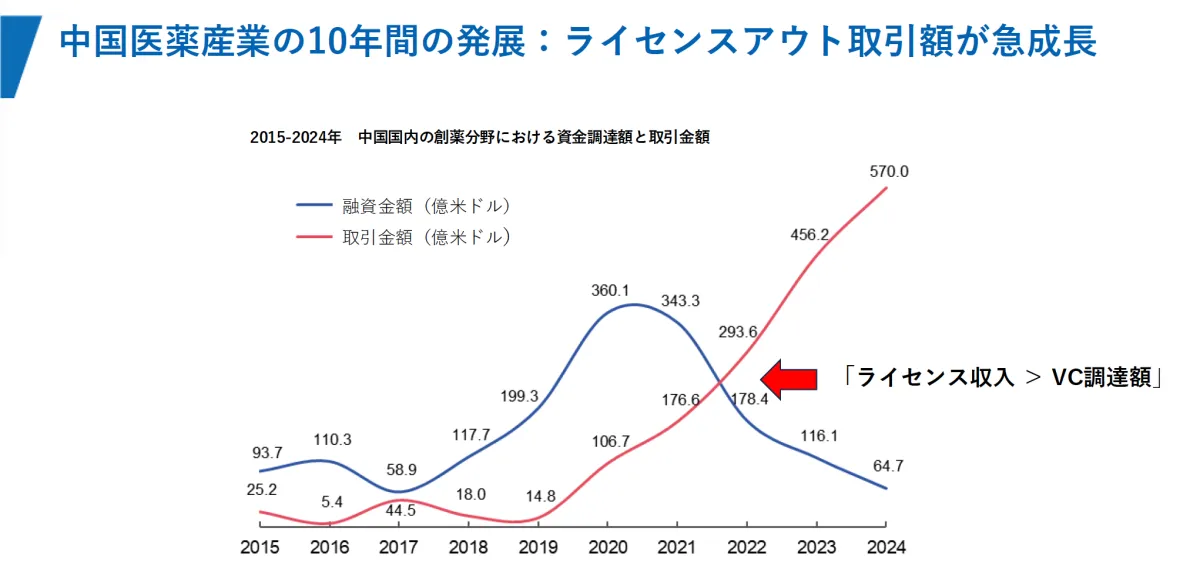
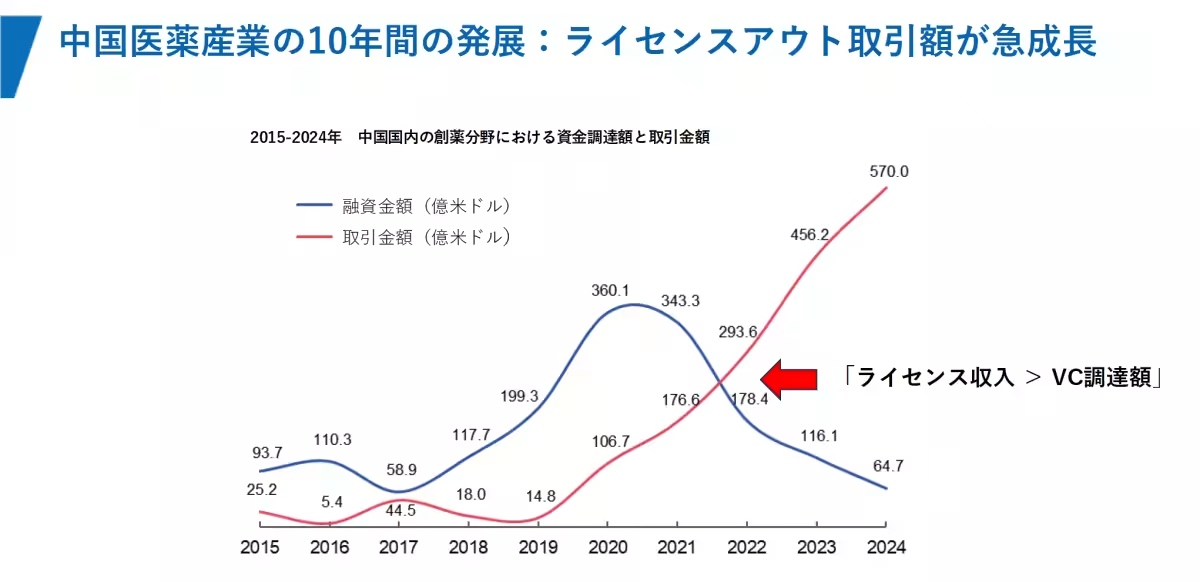
1. Transformation of Funding Structures
In 2025, the total upfront payments for license-out agreements by Chinese biotech companies reached an impressive $6.5 billion, surpassing the total amount raised through primary market investments by VC firms ($4.1 billion) during the same period. The number of annual transactions hit a record high of 157, with a cumulative deal value of $136.5 billion, establishing license-out agreements as a primary means for Chinese biotech companies to recoup investments.
2. Collaborations with Japanese Companies
A spotlight is placed on notable collaborations, such as the $11.4 billion agreement between Takeda Pharmaceutical and Innovent Biologics signed in October 2025. Astellas Pharma also engaged in a deal with a Chinese firm for the introduction of next-generation ADCs worth up to $1.34 billion. The white paper delves into the strategic backgrounds and transactions of these Japanese mega-pharma companies, offering a detailed narrative of their evolving roles in the Chinese market.
Remarks from SUGENA’s CEO, Isao Sugahara
As the CEO of SUGENA, Isao Sugahara stated, the speed and scale of research and development infrastructure in China, capable of handling everything from preclinical trials to large-scale clinical studies, has become an indispensable element of the global drug discovery ecosystem. The way Japanese pharmaceutical companies respond to innovations originating from China will play a crucial role in determining their competitiveness on a global scale over the next decade. Sugahara hopes that this report will assist companies in making informed decisions regarding these shifts.
Overview of the Report
- - Title: China Pharmaceutical Innovation White Paper 2026: The True Value of China’s Drug Discovery as Licensing Fees Surpass VC Funding
- - Publication Date: March 2026
- - Publisher: SUGENA Co., Ltd.
- - Key Contents:
- Evolution of national policies
- License-out transaction performance (2025 total: 157 deals worth $136.5 billion)
- Latest partnership examples from Japanese mega-pharma (Takeda, Astellas)
- Glossary of terms (including the 722 incident and the 4+7 system)
- - Target Audience: Business Development and Management Planning professionals in pharmaceutical and medical device companies, investors in the life sciences sector
- - Price: Free
How to Download the Report
For those interested in downloading the report for free, visit here.
About SUGENA Co., Ltd.
SUGENA is a consulting firm based in Shibuya, Tokyo, dedicated to supporting Japanese companies in cross-border open innovation. With over 34 years of experience, CEO Isao Sugahara leverages extensive overseas business experience and local networks to facilitate partnerships, from partner search to negotiation, primarily in the life sciences sector.
Location: 8-7 Daikanyamacho, Shibuya-ku, Tokyo
Contact Person: Isao Sugahara, CEO
Website: SUGENA Official Site

Topics Business Technology)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.