

Achieving Ionic Conductivity Comparable to Organic Electrolytes Using Pyrochlore-type Oxide Solid Electrolytes
Introduction
The intersection of safety and performance has become a top priority in battery technology, particularly in response to recent safety concerns surrounding lithium-ion batteries. In this context, a new development by researchers at the National Institute of Advanced Industrial Science and Technology (AIST) shines a light on the promising pyrochlore-type oxide solid electrolytes, noted for achieving ionic conductivities of 15 mS/cm. This level of conductivity is comparable to that of conventional organic electrolytes, opening new avenues for solid-state battery applications.
The Significance of Solid-State Batteries
With the rising instances of fires involving lithium-ion batteries (LIBs), there is an urgent need for safer energy storage solutions. Solid-state batteries, which utilize non-flammable inorganic electrolytes instead of flammable organic solvents, are emerging as strong candidates for next-generation batteries. Among these, oxide-based solid electrolytes are gaining attention due to their durability, thermal stability, and potential for extended lifespan.
However, there has been a significant challenge in achieving satisfactory ionic conductivity with traditional oxide solid electrolytes, which lagged behind their organic counterparts, particularly when high output is needed in applications such as electric vehicles (EVs). The breakthrough of developing oxide solid electrolytes with ionic conductivity comparable to organic electrolytes is thus crucial.
Innovative Approach to Ion Conductivity
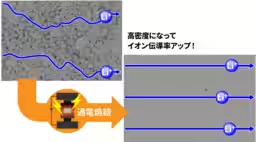
Recent studies involving the pyrochlore-type solid electrolytes have shown promising results. While conventional oxide solid electrolytes struggled with low ionic conductivity, the unique characteristics of pyrochlore compounds present a valuable opportunity for improvement. Researchers demonstrated that through fine-tuning the densification process using Spark Plasma Sintering (SPS), they could achieve a theoretical density ratio of 98%. This advancement confirmed that the pyrochlore-type solid electrolyte (Li1.25La0.58Nb2O6F) exhibited the highest ionic conductivity recorded for oxide solid electrolytes at 15 mS/cm.
The impact of this research cannot be overstated. By overcoming the challenges of densification, the study not only improves the ionic conductivity of oxide solid electrolytes but potentially accelerates the development of safe and effective solid-state batteries.
Research Context and Background
AIST has been innovating in the fields of ceramic materials for over 20 years, with a robust understanding of SPS techniques. SPS technology applies both electric current and pressure to compact materials rapidly, allowing for precise control of heating and densification, drastically reducing the time required to achieve high-density products.
Through this extensive background, AIST researchers could effectively leverage these techniques to assess and enhance the ionic conductivity of pyrochlore-type solid electrolytes.
Experimental Results and Future Directions
In laboratory tests, the pyrochlore-type solid electrolytes not only demonstrated satisfactory ionic conductivity at room temperature but were also evaluated for their performance at extreme temperatures, including as low as -100°C. This property signals the potential for applications in challenging environments, such as polar or space conditions.
Future research directions include cell design and performance evaluation of solid-state batteries using pyrochlore-type electrolytes. With efforts geared toward achieving energy densities, cycle life, and output characteristics on par with existing LIBs, the quest for creating a next-generation solid-state battery system continues.
Conclusion
The research achieved a remarkable potential for solid rechargeable batteries through the synthesis of pyrochlore-type oxide solid electrolytes. This finding represents an essential step towards the development of high-safety, high-performance solid-state batteries that combine superior ionic conductivity with the thermal and chemical stability of oxide materials. The full impact of this research will be detailed in a paper set to be published in "ACS Materials Letters" on March 11, 2026, offering a thorough account of the methods, results, and anticipated advancements in battery technology.
References
To learn more about the details of this research, refer to the publication in "ACS Materials Letters" by Yushi Fujita et al. (DOI: 10.1021/acsmaterialslett.5c01541). For additional resources, visit AIST’s official press release.



Topics Energy)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.