
ArkMS and Emuiz Forge Strategic Partnership for CRO Expansion with Focus on Safety and Quality
In a significant move to expand its Contract Research Organization (CRO) business, ArkMS, a subsidiary of the Alfresa Group, has recently established a strategic partnership with Emuiz, a leading provider in the field of safety support services for pharmaceuticals and medical devices. The announcement, made on March 31, 2026, marks a pivotal step in enhancing the quality and efficiency of medical safety evaluations and related support activities in Japan's healthcare landscape.
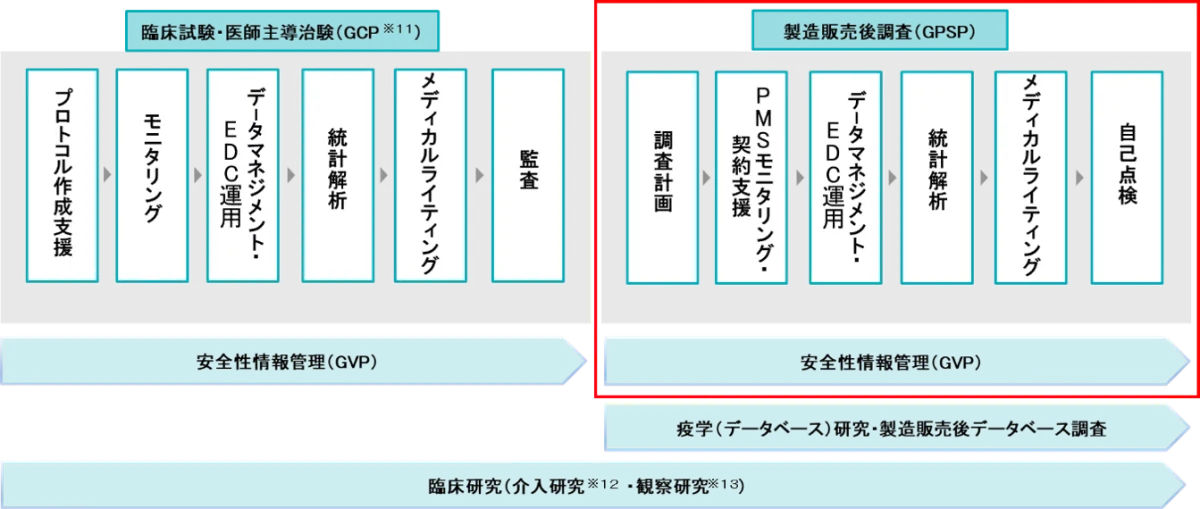
The partnership comes in the wake of evolving regulatory landscapes, particularly with guidelines issued by Japan's Ministry of Health, Labour and Welfare. These guidelines suggest that if there are no specific issues to be evaluated during approval or post-marketing phases, the effectiveness of marketed products could be assessed through literature analysis rather than solely relying on post-marketing surveillance (PMS). However, in cases where the number of clinical trial subjects is limited, the importance of PMS becomes paramount, necessitating ongoing safety evaluations and promoting appropriate use of medical products.
ArkMS is positioning itself as a pivotal player in offering tailored services that go beyond conventional post-marketing surveys, meeting the increasing demand for planning, consulting, and creating risk management plan (RMP) materials, alongside medical writing services. Through its integrated supply chain solution, known as TSCS, ArkMS aims to support emerging biotech companies' entry into the Japanese market seamlessly.
The collaboration aims to pool the capabilities of both companies, leveraging Emuiz's strengths in Good Vigilance Practice (GVP) and Good Post-marketing Study Practice (GPSP), along with their established medical writing and pharmacovigilance competencies. The two firms plan to enhance their collective offerings, fostering robust improvements in quality and proposal capabilities while expanding their sales networks to increase opportunities for both existing clients and new business engagements.
The partnership will see ArkMS and Emuiz collaboratively deliver a comprehensive array of services aimed primarily at supporting the safety of pharmaceuticals and medical devices. These services include:
1. Medical writing services
2. Planning support for PMS
3. Consulting related to Pharmacovigilance (PV) and PMS
4. Creation of materials pertaining to RMP and appropriate use information
5. Data management and statistical analysis services
6. Any other services mutually agreed upon by both companies
This joint initiative not only aims to strengthen the operational framework of both ArkMS and Emuiz in their respective domains but also supports the overarching goal of enhancing safety monitoring and efficacy evaluation processes for marketed drugs and medical devices in Japan.
While the immediate impact of this partnership on ArkMS's financial performance is expected to be minimal, it is projected to significantly enhance the company's value proposition in the mid to long term. The alignment of both organizations’ expertise signals a commitment to bolstering the CRO landscape within Japan and improving the safety and effectiveness of healthcare products available in the market. ArkMS is committed to keeping stakeholders informed of any developments related to this initiative that may arise in the future.
In conclusion, the collaboration between ArkMS and Emuiz represents a proactive step to address the evolving challenges in the pharmaceutical and medical device sectors, aiming not only to ensure regulatory compliance but to set benchmarks in quality and safety management practices for years to come.
Background and Objectives
The partnership comes in the wake of evolving regulatory landscapes, particularly with guidelines issued by Japan's Ministry of Health, Labour and Welfare. These guidelines suggest that if there are no specific issues to be evaluated during approval or post-marketing phases, the effectiveness of marketed products could be assessed through literature analysis rather than solely relying on post-marketing surveillance (PMS). However, in cases where the number of clinical trial subjects is limited, the importance of PMS becomes paramount, necessitating ongoing safety evaluations and promoting appropriate use of medical products.
ArkMS is positioning itself as a pivotal player in offering tailored services that go beyond conventional post-marketing surveys, meeting the increasing demand for planning, consulting, and creating risk management plan (RMP) materials, alongside medical writing services. Through its integrated supply chain solution, known as TSCS, ArkMS aims to support emerging biotech companies' entry into the Japanese market seamlessly.
Strategic Synergies
The collaboration aims to pool the capabilities of both companies, leveraging Emuiz's strengths in Good Vigilance Practice (GVP) and Good Post-marketing Study Practice (GPSP), along with their established medical writing and pharmacovigilance competencies. The two firms plan to enhance their collective offerings, fostering robust improvements in quality and proposal capabilities while expanding their sales networks to increase opportunities for both existing clients and new business engagements.
Services Offered Through the Partnership
The partnership will see ArkMS and Emuiz collaboratively deliver a comprehensive array of services aimed primarily at supporting the safety of pharmaceuticals and medical devices. These services include:
1. Medical writing services
2. Planning support for PMS
3. Consulting related to Pharmacovigilance (PV) and PMS
4. Creation of materials pertaining to RMP and appropriate use information
5. Data management and statistical analysis services
6. Any other services mutually agreed upon by both companies
This joint initiative not only aims to strengthen the operational framework of both ArkMS and Emuiz in their respective domains but also supports the overarching goal of enhancing safety monitoring and efficacy evaluation processes for marketed drugs and medical devices in Japan.
Future Outlook
While the immediate impact of this partnership on ArkMS's financial performance is expected to be minimal, it is projected to significantly enhance the company's value proposition in the mid to long term. The alignment of both organizations’ expertise signals a commitment to bolstering the CRO landscape within Japan and improving the safety and effectiveness of healthcare products available in the market. ArkMS is committed to keeping stakeholders informed of any developments related to this initiative that may arise in the future.
In conclusion, the collaboration between ArkMS and Emuiz represents a proactive step to address the evolving challenges in the pharmaceutical and medical device sectors, aiming not only to ensure regulatory compliance but to set benchmarks in quality and safety management practices for years to come.


Topics Health)









【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.