

AI Solution Revolutionizes SOP Management for Pharma Industry: Roketta's Latest Launch
AI Solution Revolutionizes SOP Management for Pharma Industry
On May 15, 2026, Roketta Co., Ltd., headquartered in Chiyoda, Tokyo, announced the official release of its AI service, "RakuYaku SOP Editor," aimed at aiding pharmaceutical companies in the integration and standardization of Standard Operating Procedures (SOPs). This service is a joint effort with Ono Pharmaceutical Co., Ltd., based in Chuo, Osaka.
The Challenge of Updating SOPs
In the pharmaceutical industry, maintaining SOPs is a strict requirement dictated by GMP regulations. This challenge isn’t only faced by drug manufacturers but also by medical device companies and Contract Manufacturing Organizations (CMOs) that deal with quality documentation.
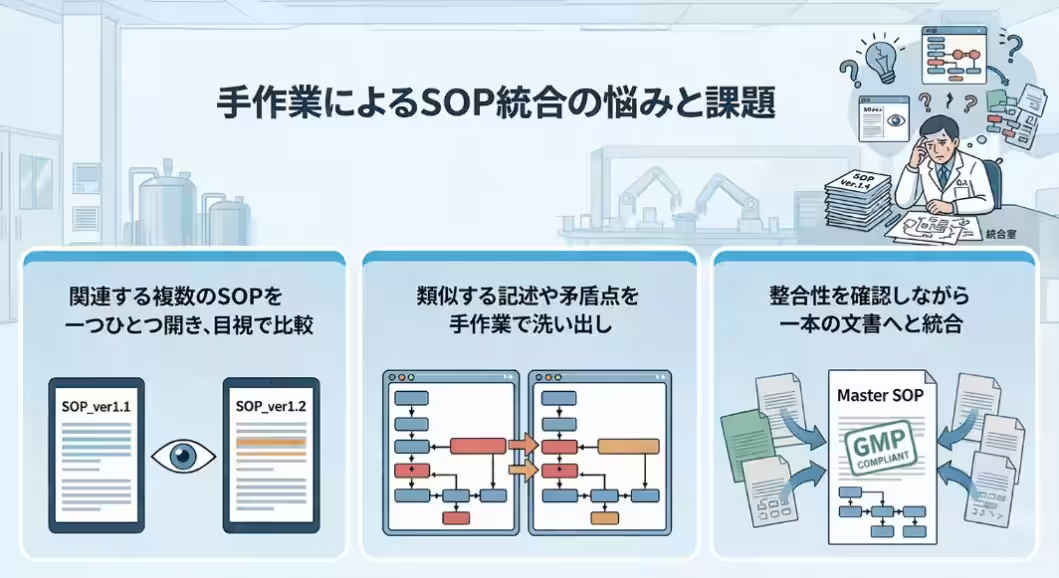
Companies often have hundreds of SOPs that are managed separately across different departments and locations. With each revision, inconsistencies in expression and structure accumulate, complicating the management processes. The necessity to integrate and update these SOPs in light of regulatory changes is a significant challenge.
Currently, personnel face an overwhelming volume of manual work when updating these documents. This labor-intensive process not only consumes a significant amount of time but also increases the risk of human error. The U.S. FDA frequently cites deficiencies in documentation as a common issue during inspections, making it imperative to enhance precision from a compliance perspective. Recognizing this urgent need, the SOP integration process is ripe for AI intervention.
Enter RakuYaku SOP Editor
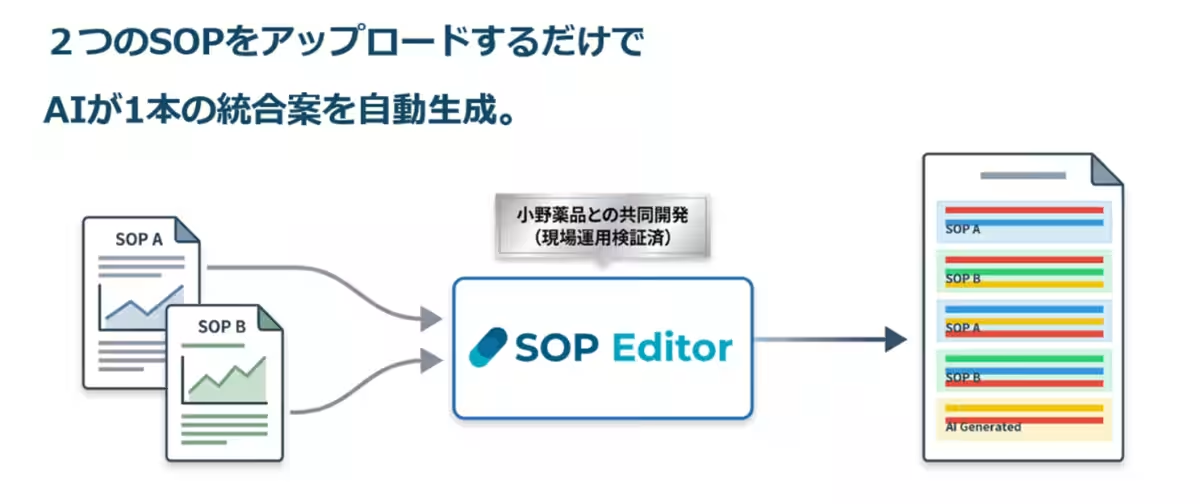
The RakuYaku SOP Editor leverages AI capabilities to reduce the burden of SOP integration.
Key Features
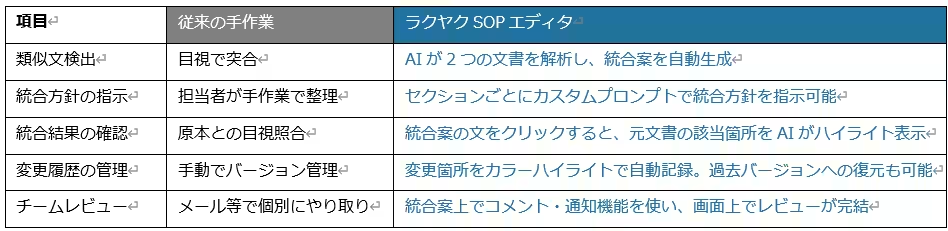
Streamlined integration
Consistency checks
Effortless customization options
Immediate SaaS-based deployment
Grounded in Real-World Insights
What sets the RakuYaku SOP Editor apart is that it was not solely developed by Roketta’s team. The project began in June 2025 through collaboration with Ono Pharmaceutical, which provided valuable insights into the challenges faced in SOP management. As regulatory timelines necessitated the creation and updating of SOPs, the burden of document integration became increasingly heavy. This input from the ground shaped the product's development. Through close collaboration with Ono’s Quality Control and Assurance teams, the practical know-how and operational workflows were directly incorporated into the development phase, resulting in a user-friendly tool tailored for real-world application.
This collaborative approach offers invaluable insights for other pharmaceutical companies facing similar challenges. The design of the RakuYaku SOP Editor addresses the structural challenges stemming from GMP regulations rather than being tailored for the specific needs of one company, allowing it to serve a broader range of industry players effectively.
Positive Reception from the Industry
Prior to its launch, the RakuYaku SOP Editor garnered considerable interest from various pharmaceutical and medical device firms, as well as Contract Research Organizations (CROs), particularly regarding its features for SOP standardization and document consistency checking. The editor not only allows for immediate SaaS deployment but also accommodates advanced customizations through a Custom Development Program (CDP), ensuring flexibility to match diverse operational workflows and existing documentation systems.
Pricing Plans
The RakuYaku SOP Editor offers three pricing tiers—Light, Standard, and Premium—tailored to the usage volume. For further details, interested parties are encouraged to reach out directly.
Future Developments
Looking ahead, the RakuYaku SOP Editor aims to expand its utility beyond individual companies and contribute to a broader “SOP Document Optimization Infrastructure” across the pharmaceutical industry. As the fifth product in the RakuYaku AI SaaS lineup, it will integrate seamlessly with existing products like RakuYaku QC Check (document consistency checking) and RakuYaku MW Editor (automated generation of clinical trial documents and applications) to strengthen an AI foundation that supports the entire drug discovery process.
Industry Insight
Yuuichi Furuya, COO of RakuYaku AI at Roketta, states, "SOPs in pharmaceutical companies are not simply completed tasks. The burden of integration arising from revisions is a structural issue common across pharmaceutical settings. I encourage other companies facing similar challenges to give it a try. You’ll experience firsthand how AI can transform the time spent on SOP comparison and integration, which will help illuminate the value of this product."
About RakuYaku AI
RakuYaku AI is a specialized generative AI SaaS solution designed specifically for the pharmaceutical industry, provided by Roketta. It supports document automation for specialized documents such as Clinical Study Reports (CSR) and package inserts, as well as ensuring document consistency checks and translations, thereby enhancing efficiency and reducing costs within pharmaceutical companies. By significantly expediting cumbersome document creation tasks, RakuYaku AI plays a critical role in accelerating drug market entry, benefiting both innovator and generic drugs alike.
About Roketta
As Japan's leading AI translation company, Roketta has helped over 6,000 clients streamline their specialized document creation through innovative AI services across various industries, including pharmaceuticals, manufacturing, law, and finance. Their flagship service features high-accuracy industry-specific translations supported by generative AI technology.
For more information on its offerings or to make an inquiry, visit Roketta's official website.
With advancements like the RakuYaku SOP Editor, Roketta is poised to lead the transformation of document management processes in the pharmaceutical sector, ensuring better compliance and operational efficiency in a highly regulated environment.



Topics Consumer Products & Retail)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.