
New Analysis Technology Enhances Quality Control in Bio-manufacturing with Cultivation Media
Introduction
In the ever-evolving landscape of biotechnology, ensuring the quality of cultivation media is paramount for successful bio-manufacturing. A new analysis technology developed by the National Institute of Advanced Industrial Science and Technology (AIST) promises to revolutionize quality control processes by allowing for rapid and efficient evaluations of the overall characteristics of culture media and supplements, rather than the traditional method of examining individual components.
The Need for Improved Quality Control
The quality of culture media and supplements is critical for the stability and reproducibility of cell and microbial cultures. However, given the complexity of these mixtures, evaluating them on a component-by-component basis is not practical due to the time and resources involved. Consequently, researchers often rely on cell culture tests, which are not only time-consuming but also subject to variability based on initial conditions and operator expertise. This leads to inconsistencies and challenges in reproducing results.
Development of New Analytical Technology
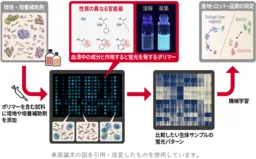
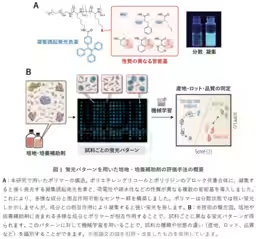
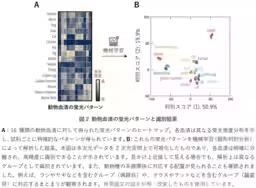
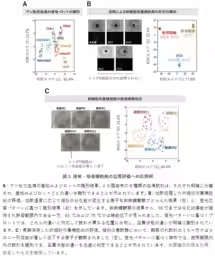
AIST's researchers, including Shunsuke Tomita, Kumi Morikawa, and others, have innovated a new analysis technique that captures the overall characteristics of cultivation materials through fluorescence patterns generated by synthetic polymer probes. By processing these patterns with data analysis methods such as machine learning, this groundbreaking technology enables high-precision detection of subtle variations in formulation and condition.
This technique is particularly noteworthy as it can identify differences in quality among various batches of culture media and supplements, including fetal bovine serum and synthetic adjuncts used for stem cell culture, without relying on traditional analysis methods or cell culture testing.
Societal Implications
The advancement in biotechnology is reshaping industries related to drug discovery, regenerative medicine, and bio-manufacturing, which are crucial for creating a sustainable society. As the demand for high-quality bio-manufactured products rises, this new quality evaluation method addresses important regulatory and production challenges stemming from the diverse compositions of culture media and supplements. Variability in these materials has the potential to create significant disruptions in production efficiency and product quality.
For instance, fetal bovine serum, a commonly used supplement, suffers from batch-to-batch variability and quality degradation due to storage conditions. Addressing the specific contributions of different components to the overall cultivation function has historically been challenging. This new approach aims to streamline quality assessment and enhance consistency across production processes, thereby mitigating the risks of manufacturing-related issues.
Research Process and Findings
The groundwork for this technology lays in AIST's previous work on the
Topics Consumer Products & Retail)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.