
Hitachi Introduces Innovative Seamless Integration for Regenerative Medicine Value Chain Management
Hitachi's Groundbreaking Approach to Regenerative Medicine Management
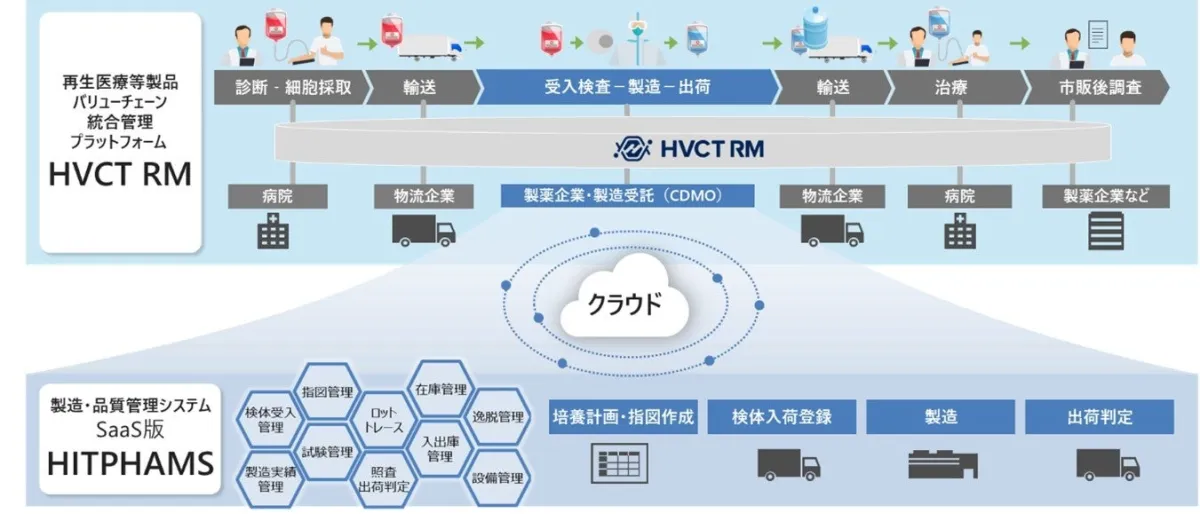
In a significant leap for the field of regenerative medicine, Hitachi has recently unveiled an innovative feature for its comprehensive management platform known as the Hitachi Value Chain Traceability Service for Regenerative Medicine (HVCT RM). This feature seamlessly integrates data from the subscription-based manufacturing and quality management system, HITPHAMS, specifically designed for the pharmaceutical and medical device manufacturing sectors. By combining these two advanced Lumada solutions, Hitachi aims to streamline various processes engaged in the management of biopharmaceuticals throughout the entire supply chain, which includes medical institutions, logistics companies, pharmaceutical firms, and contract development and manufacturing organizations (CDMOs).
The new integration feature automatically connects tracing information and production data such as manufacturing orders and shipment evaluations, significantly reducing the workload that was previously reliant on human input. By enabling real-time sharing of information on a singular system, stakeholders can minimize risks associated with product misidentification while promoting prompt decision-making and scheduling adjustments. This approach not only facilitates more efficient operations but also helps prevent potential losses in treatment opportunities for patients.
Innovations Focused on Data Integrity
One of the standout aspects of this new integration is its commitment to ensuring data integrity. Patients’ sensitive information and the confidential data of organizations are safeguarded through advanced permission management systems, allowing for precise control over who can view what information and when. This security measure is vital in preserving patient privacy while ensuring that the necessary data is shared with appropriate stakeholders involved in the manufacturing and treatment processes.
Hitachi’s HITPHAMS system is recognized as one of the top-quality management systems in Japan for the pharmaceutical and medical device industries, emphasizing swift installation and operation without overwhelming clients with burdensome operations. The HVCT RM platform has successfully been adopted by various pharmaceutical manufacturers, wholesalers, and biotech ventures across the nation. By continuing to innovate within IT and Operational Technology (OT) systems, Hitachi aims to address a broad spectrum of challenges present in the regenerative medicine value chain.
Showcasing Innovations at Nakanoshima Qross
To exhibit its cutting-edge technologies, Hitachi provides showcases of its solutions at the Nakanoshima Qross in Osaka and the Regenerative Medicine Innovation Center in Tokyo, allowing stakeholders to witness firsthand the advancements in regenerative medicine solutions. The company is particularly focused on implementing next-generation solutions termed “HMAX Industry,” which blends domain knowledge and leading-edge AI technologies to innovate frontline operations in manufacturing and nurturing.
This ongoing commitment to technological advancement reflects Hitachi's holistic view of Health Care as a strategic sector deserving attention and investment. With the accelerated demand for regenerative medicines, propelled by aging populations and advancements in healthcare technologies, the company recognizes the urgent need for enhanced traceability and management throughout the supply chain of regenerative products.
Reducing Errors and Streamlining Processes
As the integration feature comes online, the reliance on manual data entry is significantly diminished. Previously, stakeholders engaged in this complex value chain faced cumbersome processes often characterized by human error through data duplication or entry mistakes. By automatically transferring essential data such as manufacturing progress and testing results from HITPHAMS to HVCT RM, the likelihood of errors is drastically reduced. Shared patient management IDs provide further assurances against product misidentification across the board.
Furthermore, the functionality of the new feature means that stakeholders can visualize shipping, manufacturing, and treatment statuses from a single platform. This streamlined view mitigates inquiry workloads, promotes efficient schedules, and addresses manufacturing delays more adeptly to reduce the chance of missing critical treatment windows for patients.
Conclusion
As the global landscape of regenerative medicine evolves, Hitachi is at the forefront with innovative solutions aimed at enhancing efficiency, security, and reliability in medical product management. With its dual systems working in concert, Hitachi is set to optimize the complex web of interactions between medical institutions, suppliers, and manufacturers. The future likely holds expanded reach into markets across North America and Asia, positioning Hitachi as a leading figure in the increasingly vital field of regenerative medicine.
With their commitment to societal innovation and harmonizing technology for improved healthcare, Hitachi remains dedicated to addressing real-world challenges and providing solutions that may redefine the future of medical treatments.
Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.