
Sino Biological Accelerates Vaccine Innovation for 2026-2027 Northern Hemisphere Influenza Strains
Advancing Vaccine Development Amid Rising Influenza Threats
In response to the World Health Organization's (WHO) latest guidance, Sino Biological, Inc. is taking proactive measures to enhance the development of influenza vaccines for the upcoming Northern Hemisphere season from 2026 to 2027. With the emergence of specific viral strains that pose a significant public health challenge, this initiative is timely and crucial.
WHO Updates on Influenza Vaccine Composition
On March 3, 2026, the WHO announced critical updates that reflect the evolving landscape of influenza strains. Central to these recommendations is the identification of the A(H3N2) subclade K, also known as J.2.4.1, which has rapidly become the predominant type of influenza A seen globally since its discovery in August 2025. Notably, this strain features mutations, specifically T135K and S144N, that enhance its ability to evade immune responses, necessitating more robust vaccine strategies.
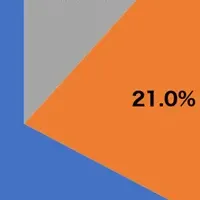
As part of its response, WHO has designated A/Darwin/1454/2025 as the reference virus for the H3N2 component, while also updating the H1N1 component to A/Missouri/11/2025. Additionally, there is a notable increase in B/Victoria lineage strains, rising from 6% to over 20% in regions such as Hong Kong SAR and the United States, leading the WHO to recommend strains B/Tokyo/EIS13-175/2025 and B/Pennsylvania/14/2025.
Sino Biological's Role in Influenza Vaccine Research
To align with these WHO recommendations, Sino Biological is launching a wide array of recombinant antigens targeted for the upcoming influenza vaccine strains. This includes vital components such as A/Missouri/11/2025 (H1N1) HA Trimer, NA, and NP proteins, which are instrumental for effective vaccine formulation. Dr. Rob Burgess, Chief Business Officer at Sino Biological US, emphasized the company's commitment to equipping the scientific community with high-quality tools that are essential for advancing vaccine research and development.
"Our mission is to provide the scientific community with the highest quality tools as rapidly as possible when infectious disease evolution threatens global health preparedness," Dr. Burgess noted. The company’s innovative research platforms and proprietary quality assurance systems play significant roles in ensuring the efficacy and reliability of its vaccine research products.
Sino Biological: A Leader in Biotechnology
Sino Biological has established itself as a global leader in the biotechnology realm, specializing in high-quality recombinant proteins and antibodies. The company serves researchers in over 90 countries, supporting various domains such as basic research, drug discovery, vaccine development, and diagnostics. Sino Biological's commitment to quality and innovation makes it a pivotal player in the fight against infectious diseases.
As we anticipate the flu season, the proactive efforts of organizations like Sino Biological will be crucial in preparing effective vaccines that could save countless lives. The integration of advanced biotechnology with timely responses to public health challenges is essential to safeguard global health in an unpredictable environment.
In conclusion, as the WHO navigates the complexities of influenza strain management, Sino Biological stands ready with its newly launched antigens to foster advancement in vaccine efficacy and development, showcasing the importance of rapid response mechanisms in public health initiatives.
For further inquiries or partnership opportunities, please reach out to Sino Biological, Inc. through their website or contact information provided in the press release.
Topics Health)









【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.