
New e-learning Course on Medical Device Industry Basics Launched by Nihon IR
Introduction to the New Course on Medical Devices
Nihon IR, a prominent player in the educational sector for manufacturing industries, has recently expanded its popular e-learning series, Tech e-L, by introducing a new course titled 'Basics of the Medical Device Industry'. This comprehensive program is designed to equip professionals—especially newcomers and recent graduates—with essential knowledge about medical devices, crucial for ensuring safety and compliance in a highly regulated industry.
Course Overview
The new course is structured to cover critical areas including the definition and classification of medical devices, relevant regulatory frameworks such as Good Practice (GxP) and Quality Management Systems (QMS) regulations, safety measures, and international standards. Additionally, it addresses the various roles and organizations within this industry and insights into the medical device market.
Target Audience
This e-learning course is ideal for:
- - New hires in the medical device sector.
- - Young professionals or those with limited experience in the field.
- - Individuals working with medical devices in any capacity.
Importance of Regulatory Knowledge in Medical Devices
Given that medical devices directly impact the safety of patients, the industry is governed by stringent legal standards, which cannot be ignored by professionals. Understanding these regulations is paramount—'I didn’t know the law' is not a valid excuse in this context. Moreover, the medical device sector often witnesses rapid product life cycles, underscoring the need for continual learning and knowledge updates.
What Will You Learn?
In this course, learners will begin with a thorough understanding of what constitutes a medical device and how they are classified. The curriculum will delve into critical regulations such as GxP and QMS, emphasizing safety protocols and international guidelines. Ultimately, participants will grasp how medical devices interact with society, exploring industry roles, associated organizations, and market dynamics.
Main Course Topics
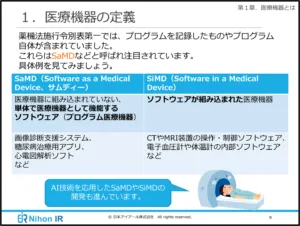
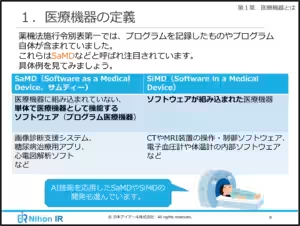
1. Definition and Classification:
- Gain clarity on what medical devices are and their various categories.
2. Information and Regulations Compliance:
- Understand the GxP and QMS regulations, safety measures, and global standards.
3. The Medical Device Ecosystem:
- Discover related professions, organizations, and overall market context.
Pricing Information
For corporate clients looking to enroll multiple participants, the course is available at a monthly fee starting from 9,800 yen per company, which includes management features for participants. The initial setup fee begins at 45,000 yen.
Conclusion
Nihon IR’s mission is to create educational environments where professionals can learn essential knowledge at the right time—supporting workplace education and development. This addition to the Tech e-L series highlights the company’s commitment to advancing education in the manufacturing sector, particularly in the context of medical devices that are crucial to human safety and health.
For more details on the course, please visit: Basic Medical Device Industry Course
And for an overview of Tech e-L, check out: Tech e-L Overview
Explore the innovative solutions offered by Nihon IR, which has over 50 years of experience in patent and intellectual property solutions, technical information analysis, and educational services in the manufacturing industry. Visit their official website at Nihon IR.


Topics Consumer Technology)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.