

Shionogi Healthcare Unveils New Cough Suppressant Tablet for Ages 8 and Up
Shionogi Healthcare Launches New Cough Suppressant
Shionogi Healthcare, headquartered in Osaka, has announced the release of ASVERIN® Cough Suppressant Pro20, a new medication specifically formulated for individuals aged eight and older. Officially launching on March 5, 2026, this product contains the same active ingredients in the same quantities as its prescription counterpart, ensuring a high level of efficacy. The new cough suppressant will be available OTC (over-the-counter) at pharmacies and drugstores across Japan.
Background of the Launch
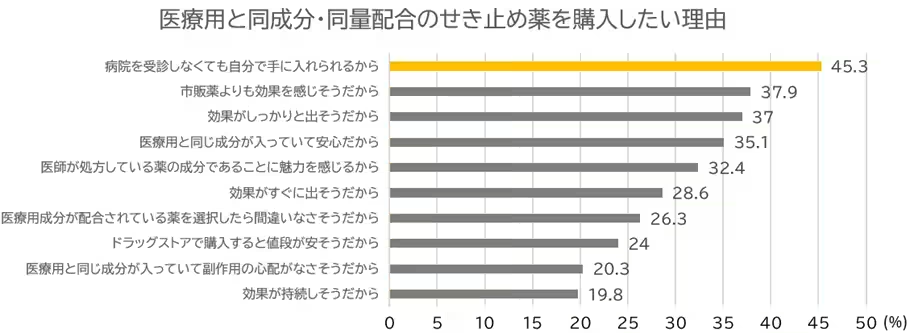
In recent years, there has been a significant increase in health-conscious consumers who are adopting the practice of self-medication for mild health issues. This trend has led to the growing use of OTC medicines as a common choice for everyday health management. However, within the expectations of a safe and effective cough suppression market, many consumers are increasingly aware of the regulatory classifications surrounding these products. According to internal surveys by Shionogi, around 69% of respondents expressed a willingness to use cough suppressants that contain the same active ingredients as their prescription versions, primarily because these could be obtained without visiting a doctor's office.
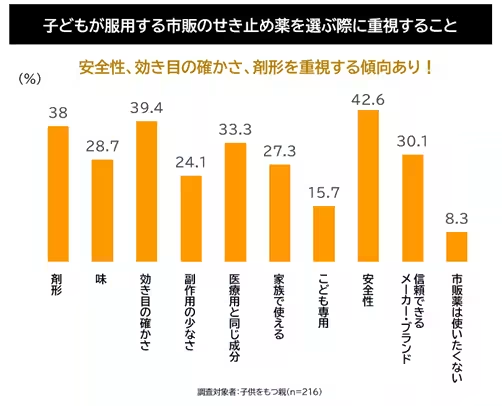
Additionally, Shionogi's research highlighted that parents of young children prioritize safety, effectiveness, and formulation when selecting cough medications. This underscores a notable demand for effective self-care options that can be directly managed at home. In response to this market environment and consumer needs, Shionogi has positioned ASVERIN® Cough Suppressant Pro20 as an ideal solution.
Product Features
- - Equivalence to Prescription Strength
- - Available for Ages 8 and Above
- - Non-Narcotic and Safe
Product Overview
The launch of ASVERIN® Cough Suppressant Pro20 marks the addition of the fourth cough relief product to the Pro Series by Shionogi Healthcare, which aims to support self-medication practices across various demographic groups. The Pro Series is part of Shionogi’s initiative to deliver health care value to the community, ensuring people can obtain the care they need effectively and safely.
Shionogi is committed to transforming self-care into a familiar and practical concept and believes this new product will significantly enhance the self-care landscape. With a firm focus on research and development, Shionogi Healthcare aims to continue providing innovative health solutions that respond to the evolving needs of patients and caregivers alike.
For more information on the product, consult the official sites of Shionogi: Shionogi Healthcare and Nipro Co..
Conclusion
The release of ASVERIN® Cough Suppressant Pro20 not only signifies Shionogi's commitment to innovation in health care but also emphasizes the importance of enabling individuals and families to manage their health proactively and effectively. As the landscape of over-the-counter medications continues to evolve, Shionogi Healthcare stands at the forefront, ready to meet the increasing demands of self-care.





Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.