
Medmain Develops AI for Automated Measurement of Ki-67 Positive Cell Rates in Digital Pathology
Medmain Develops Innovative AI for Ki-67 Measurement
Medmain Inc., headquartered in Fukuoka, Japan, has made significant strides in digital pathology by creating an AI system capable of automatically calculating the positivity rate of Ki-67 protein through immunohistochemical digital slides. Published in the esteemed journal Diagnostics, this breakthrough promises to enhance the efficiency and accuracy of pathological assessments.
Overview of the Research
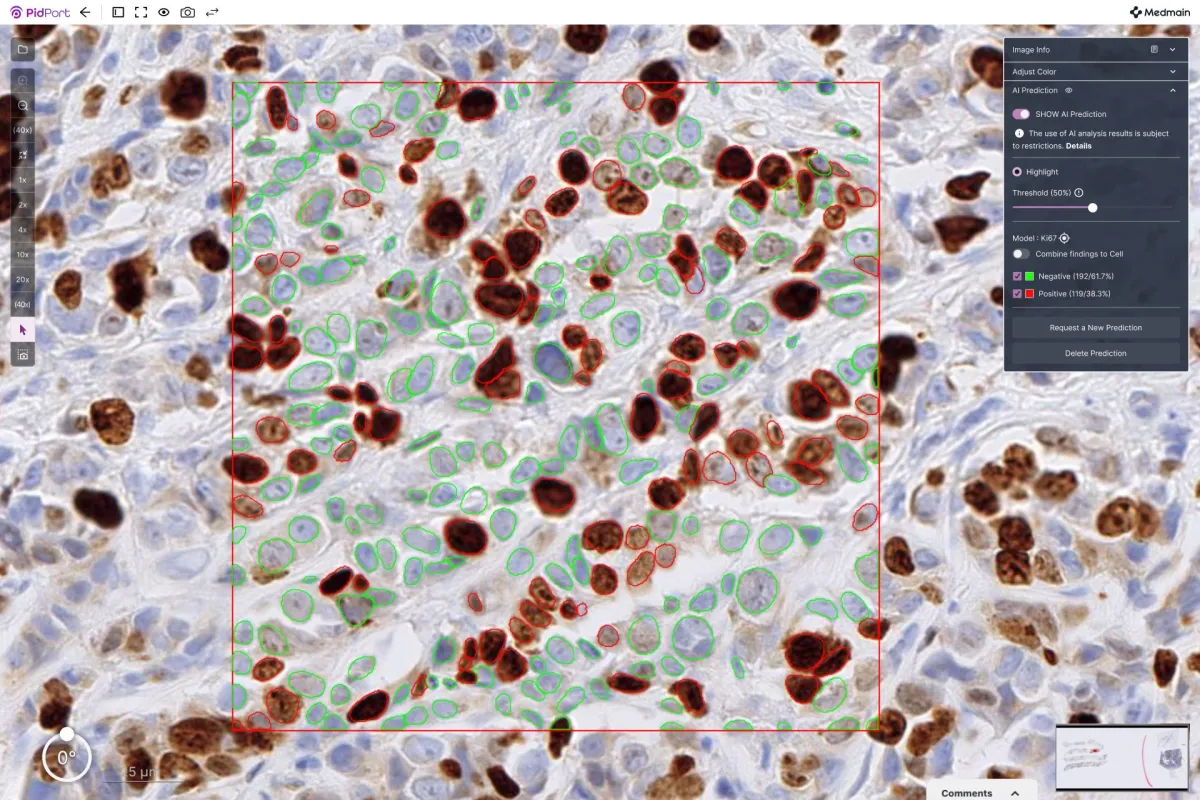
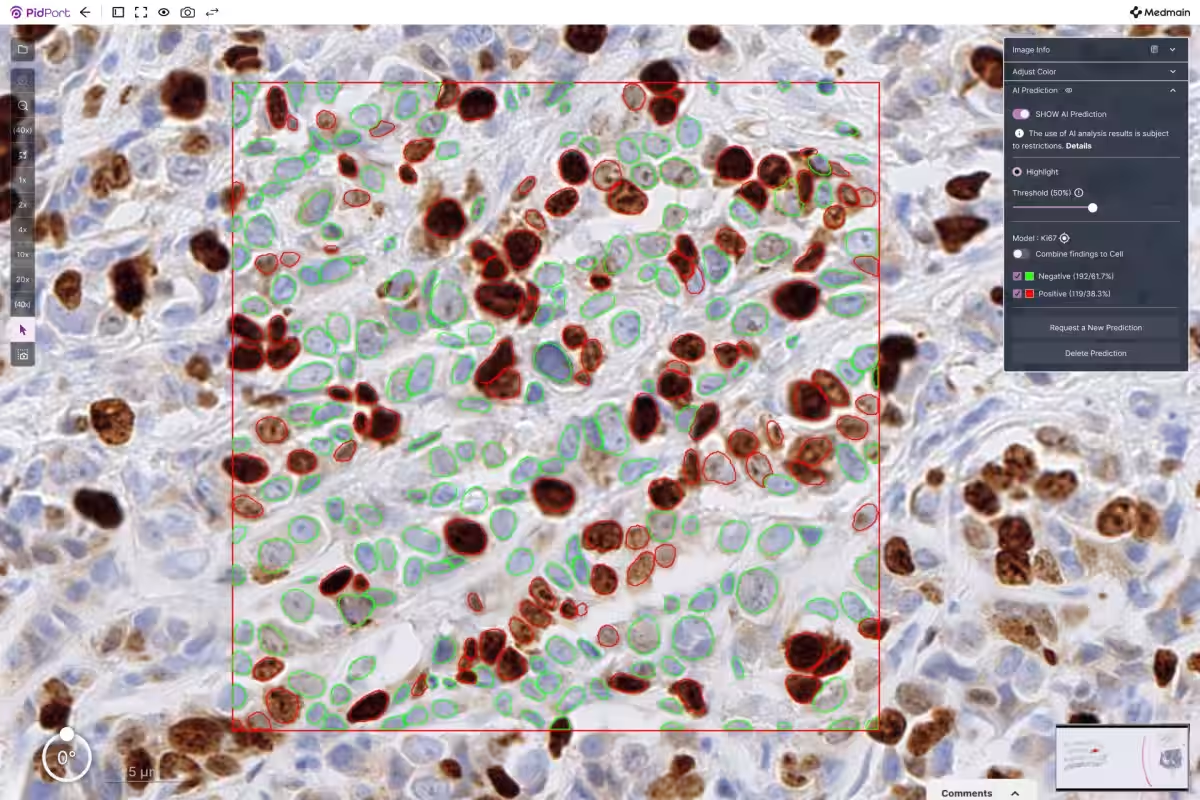
The developed AI effectively recognizes and classifies Ki-67 positive and negative cells on Whole-Slide Images (WSI), ultimately calculating the Labeling Index (LI). This study demonstrated exceptional classification performance, yielding a high correlation with evaluations performed by pathologists.
Background of the Study
Ki-67 protein (MKI67) is a well-known cell proliferation marker. It is uniquely expressed during all phases of cell division (G1, S, G2, M) but is absent during the resting phase (G0). Pathologists utilize immunohistochemistry (IHC) to visualize and assess Ki-67 expression, making it a critical indicator of tumor proliferation in cancers such as breast cancer and neuroendocrine tumors.
Historically, pathologists have counted thousands of cells manually to determine the Ki-67 positivity rate in cancer samples. However, this process can be labor-intensive and time-consuming, highlighting the pressing need for objective and efficient measurement software.
Medmain's research sought to address this need by developing a model capable of automating the identification of Ki-67 positive cells and calculating their positivity rate.
Research Methodology
The researchers digitized 320 Ki-67 immunohistochemical stained samples from various cancers, including breast cancer, sourced from domestic medical facilities. Pathologists annotated the presence of Ki-67 positive and negative cells, which served as the training data for the AI model.
The AI's initial step involved the detection of cell nuclei, followed by the classification of these cells into positive and negative categories using the trained model. Consequently, the algorithm can now automatically count Ki-67 positive and negative cells, calculating the Labeling Index efficiently.
An additional feature allows pathologists to specify regions of interest (ROI) in the Whole-Slide Images, enabling the AI to detect all cells within that designated area and perform classification and rate calculation accordingly.
Results of the Study
The classification performance for Ki-67 positive cells achieved an impressive ROC-AUC score of 0.981. Moreover, the positivity rates calculated by the AI showed a significant correlation with evaluations done by pathologists on the same images. Thus, the AI model has been confirmed as a valuable tool to assist medical professionals in evaluating Ki-67 positivity rates.
Future Prospects
While this study focused on Ki-67, the methodologies developed could potentially be adapted for other proteins exhibiting positive signals within the nucleus, such as P53, under immunohistochemical analysis. Future research will not only aim at various nuclear proteins but also extend to other cellular localizations, including membrane proteins, facilitating objective and quantitative evaluation across a wider protein spectrum.
Publication Details
- - Title: Automated Assessment of Ki-67 Labeling Index Using Cell-Level Detection and Classification in Whole-Slide Images
- - Journal: Diagnostics
- - DOI: Link to Article
- - URL: Read More
This research was conducted with the support of the Fukuoka Prefecture Innovative Medical Device Research and Development Subsidy.
About Medmain Inc.
Founded in January 2018, Medmain Inc. focuses on the planning, development, and operation of healthcare software and cloud services. The company operates various offices in Tokyo, Fukuoka, and Silicon Valley, providing innovative solutions in the medical technology landscape.
For more information, visit Medmain's Corporate Site or explore Medmain's Product Services.

Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.