
Toray Research Center Expands Support for Antibody Drug Quality Evaluation
Toray Research Center's Commitment to Antibody Drug Quality
Toray Research Center (TRC), located in the heart of Tokyo, Japan, has made significant strides in addressing the growing complexities in the development of antibody drugs. As the pharmaceutical landscape shifts increasingly towards biopharmaceuticals, especially monoclonal antibodies and antibody-drug conjugates (ADCs), TRC aims to tackle the pressing issue of quality evaluation which has become a critical bottleneck in medication implementation.
Addressing Ongoing Challenges
Recently, the demand for innovative therapies focusing on cancer and autoimmune disorders has surged. However, as the molecular structures of these antibody drugs become more complex, ensuring rigorous quality evaluations and stability testing has become essential in the drug development process. TRC has thus strengthened its evaluation framework to encompass the entire lifecycle of antibody drugs, from development to post-market activities.
With over 25 years of analytical experience in antibody drug development, TRC is now building a comprehensive system that aligns with Good Manufacturing Practice (GMP) regulations. This system supports the complete spectrum of analytical requirements, from structural analysis during the development phase to stability testing after launch, guaranteeing consistency in quality.
Innovative Analytical Technologies
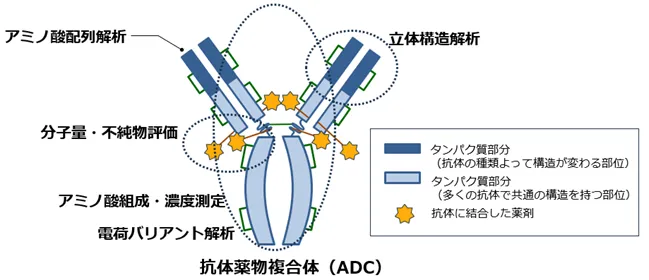
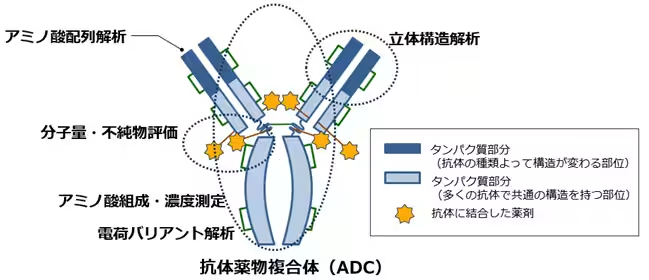
To enhance their capabilities, TRC recently integrated a new Capillary Isoelectric Focusing (cIEF) device into its GMP-compliant stability testing regimen. This state-of-the-art technology can detect even the slightest charge differences in complex antibody structures with high precision, allowing for improved structural analysis of ADCs, which have been gaining traction as next-generation therapies. This enhancement significantly boosts TRC's ability to respond to the growing demand for precise analyses of ADCs and similar complex formulations.
Through these advancements, TRC is establishing a one-stop solution for antibody drug development and quality assurance, promoting both expediency and reliability in the pharmaceutical industry. They are committed to facilitating swift social implementation of groundbreaking drugs by integrating development speed with robust quality standards.
The Landscape of Antibody Drug Development
The evolution of drug modalities has led to explosive growth in the market for biopharmaceuticals. New approaches, such as gene therapies and mRNA vaccines, continue to emerge, broadening the market further. Particularly, antibody drugs are experiencing heightened demand due to their versatile application across a range of illnesses—including various forms of cancer and autoimmune diseases.
For the development and manufacturing of these cutting-edge pharmaceuticals, reliable analytical technologies and a rigorous quality evaluation framework are not just advantageous but necessary. Each antibody drug consists of large and intricate proteins, leading to subtle structural differences influenced by post-translational modifications. This complexity necessitates multifaceted and precise structural assessments, which TRC is poised to deliver through its well-developed GMP-compliant systems.
Continuous Advancement in Analytical Techniques
Despite having already established robust systems under GMP, TRC continues to innovate. The new cIEF apparatus is capable of high-resolution analysis, allowing researchers to discern minor charge differentials, making it invaluable for the detailed characterization needed in ADCs and other sophisticated antibody constructs. This initiative ensures TRC's commitment to providing stable, high-quality, and reliable data steadfastly.
Future Directions
Going forward, TRC aims to elevate its analytical technologies further, positioning itself as a pivotal analysis partner in the pharmaceutical development realm. They strive to support every stage of drug development while resolving their clients' specific challenges through enhanced capabilities and service expansion. By continuously integrating the latest analytical advancements, TRC is set to play a vital role in accelerating the development of pharmaceuticals, enabling early societal access to innovative therapies.
In conclusion, TRC's forward-thinking approach in expanding its analytical services signifies a promising future for antibody drug evaluations and overall pharmaceutical development, ensuring that complexities of modern medicine meet their expected efficacy and safety standards.

Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.