

Rosetta and Compound Safety Research Institute Launchs AI-Driven CSR Editor on April 16 to Transform Clinical Trials
Revolutionizing Clinical Trials with the New CSR Editor
On April 16, 2026, a collaboration between Rosetta, a leading AI translation provider, and the Compound Safety Research Institute is set to transform the landscape of clinical trials in Japan. The new CSR (BE) Editor, a cutting-edge tool developed for creating Clinical Study Reports (CSR) for biological equivalence testing, will officially commence operations. This initiative aims to enhance the accuracy and efficiency of the CSR preparation process, which has traditionally involved extensive manual efforts prone to errors.
The Need for Transformation
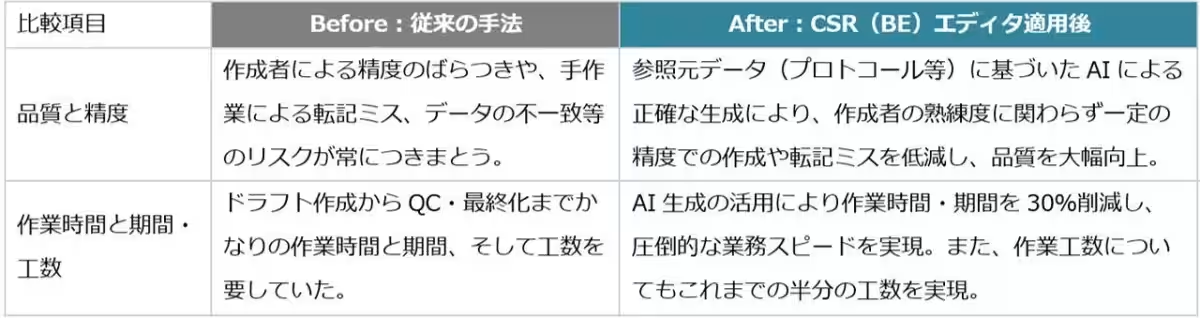
The creation of CSRs in clinical trials typically results in documents spanning more than 1,000 pages. This volume poses significant challenges, including manual transcription errors and inconsistencies in data. To combat these issues, Rosetta has utilized generative AI technology to automate CSR generation, ensuring a high accuracy rate; approximately 97% of project details can be applied with minimal revisions.
In collaboration with the National Cancer Center Hospital, the entity has validated the effectiveness of its AI capabilities, proving the potential for widespread application within the field of pharmaceutical research and development.
Expanding AI Capabilities
By leveraging its advanced AI foundation, Rosetta has expanded the CSR Editor's functionalities to cater specifically to the biological equivalence testing domain. Partnering with the Compound Safety Research Institute, they have developed a specialized CSR editor that meets the rigorous standards necessary for practical deployment, set to be operational from April 16, 2026.
Transformative Changes in Reporting
The introduction of this innovative tool will dramatically shift the CSR preparation process in biological equivalence trials. The CSR Editor provides:
1. Automated Document Generation: Scripts can now generate extensive reports with predefined templates, saving time and resources.
2. Quality Assurance: AI-driven checks ensure that all documents maintain high accuracy and consistency, significantly reducing the risk of human error.
3. Cost Efficiency: Enhanced speed in the report generation translates into substantial cost savings for pharmaceutical companies involved in clinical trial operations.
Future Prospects
As the CSR Editor launches, it is expected to demonstrate its superior cost performance and time-saving capabilities in the upcoming contracted tests for this fiscal period. Rosetta is committed to advancing Rakuyaku AI, its flagship generative AI solution, which serves the pharmaceutical sector with robust functionalities for automating and streamlining document creation. This endeavor is vital for accelerating drug development processes and fostering growth within the pharmaceutical industry.
About the Compound Safety Research Institute
The Compound Safety Research Institute offers a full spectrum of services, focusing on both non-clinical and clinical safety trials for a variety of fields, including pharmaceuticals, medical devices, and regenerative medicine. Their mission is to provide seamless support from preclinical to clinical testing phases. Services include:
1. Non-Clinical Trials: Safety testing for pharmaceuticals, medical devices, agrochemicals, food, and cosmetics.
2. Clinical Trials: Support for various clinical studies, including monitoring, auditing, statistical analysis, and documentation.
For more information, visit Compound Safety Research Institute.
Insights into Rakuyaku AI
Rakuyaku AI, developed by Rosetta, is a generative AI SaaS solution specifically designed for the pharmaceutical industry. It automates the production of specialized documents like CSRs and product inserts, performing consistency checks and translations to streamline operational efficiency and reduce costs. By significantly accelerating the documentation process, it aids in the swift market entry of both original and generic pharmaceuticals. Explore more about Rakuyaku AI at Rakuyaku AI Product Site.
Conclusion
This collaboration marks a substantial leap towards technological advancement in the realm of clinical trials. With potent tools like the CSR Editor and Rakuyaku AI, Rosetta and the Compound Safety Research Institute are poised to lead pharmaceutical innovation into a new era where efficiency and accuracy are paramount.

Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.