
Revealing the Mechanism of High-Temperature Active Enzymes for Plastic Recycling
Understanding High-Temperature Active Enzymes
Introduction
Recently, the potential of enzymatic recycling technology has caught attention, especially regarding the breakdown and reuse of polyethylene terephthalate (PET), a common type of plastic. While enzymes are generally sensitive to heat, a specific enzyme known as cutinase derived from the microorganism Chaetomium thermophilum has shown promising capabilities to function under high-temperature conditions, thus enhancing the efficiency of PET decomposition. This article delves into the detailed findings from the research conducted by the team at Tokyo University of Science, led by Professor Tatsuya Nishino and others.
Research Motivation
PET is widely used in products such as plastic bottles. It has the property of becoming softer and easier to decompose as the temperature rises, especially around 70°C. However, standard enzymes tend to denature (lose their functional shape) at high temperatures, limiting their efficacy in real-world applications. This led researchers to seek out heat-stable enzymes that retain functionality even in challenging conditions.
Study Overview
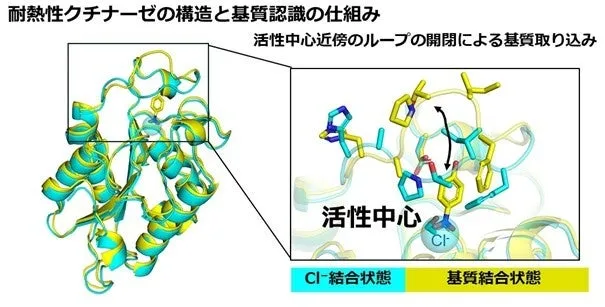
The study utilized X-ray crystal structure analysis and Differential Scanning Calorimetry (DSC) to investigate the structural and thermal stability of the cutinase enzyme. One significant finding revealed that the enzyme retains a stable overall structure while demonstrating flexibility in specific regions near its active site. This feature is particularly crucial for enhanced molecular recognition and activity.
The research group observed that a lid-like structure near the active site of the enzyme changes configuration based on the substrate it binds. In the case of the wild-type enzyme, chloride ions were found near the active site; however, substituting these ions with p-nitrophenol led to significant structural changes in the lid, facilitating the enzyme's catalytic abilities.
Detailed Findings
Using DSC, the team identified that the enzyme exhibited phase transitions around 66.4°C and 69.5°C, implying that the enzyme alters its structure not in a single step but gradually, which suggests the presence of multiple thermally stable regions within the enzyme. The X-ray crystal structure analysis illustrated that the enzyme maintains a consistent fundamental structure in the absence of substrate. Specifically, the lid was found to be in a closed position and was not immediately ready for reaction.
Upon introducing the synthetic compound p-nitrophenol, it displaced chloride ions at the active site, prompting the lid to open. This observation indicated that the enzyme could dynamically adapt its structure based on the molecule it interacts with, a crucial characteristic for enzymatic reactions.
Implications for PET Recycling
This research presents crucial insights into designing enzymes that combine both heat resistance and functionality, which could vastly improve environmental recycling methods for plastics. The findings further emphasized the necessity for continued research on the structural dynamics of not only this enzyme but also regarding the binding mechanisms of PET to fully understand the enzymatic breakdown processes involved.
While this study lays the groundwork for future advancements in PET recycling, further analysis is needed to detail the interaction between PET and the enzyme, as well as the subsequent reactions that lead to its degradation.
Conclusion
The work done by Professor Nishino and his team positions the cutinase enzyme as a significant player in enzymatic recycling technology. It underscores the importance of structural biology in addressing critical environmental challenges, specifically in developing practical methods for recycling plastics. Their research has been published in the academic journal "Crystals" and marks a vital step towards more sustainable practices in waste management and environmental conservation.
Future Directions
The findings encourage further investigations into engineering even more resilient and efficient enzymes that can decompose various types of plastics, particularly under varying environmental conditions. As the demand for sustainable solutions increases, researches like this align well with global efforts to mitigate plastic pollution and promote recycling technologies.
Topics Environment)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.