
Sino Biological Unveils Cutting-Edge Influenza Vaccine Antigens for 2026-2027
Advances in Influenza Vaccine Development
Sino Biological has made significant strides in the development of influenza vaccine antigens, providing essential components for the upcoming northern hemisphere strains for 2026-2027. This announcement comes on the heels of new recommendations from the World Health Organization (WHO), which emphasizes the need for updated vaccines due to the evolving landscape of influenza viruses.
WHO Recommendations Highlight New Challenges
The WHO recently outlined its recommendations for the composition of influenza vaccines for the northern hemisphere, pointing out the continuing spread of subclade K of the A(H3N2) strain, alongside emerging variants from the B/Victoria lineage. The increasing prevalence of these strains calls for timely action in vaccine formulation to ensure effective protection against the flu in the coming seasons.
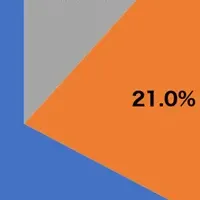
The emergence of the K subclade (J.2.4.1), which showcases mutations T135K and S144N, has led to a notable increase in its ability to evade immunity. This evolution prompted the WHO to identify A/Darwin/1454/2025 as the new reference virus for H3N2. In addition to this, the H1N1 component has been updated to A/Missouri/11/2025. Moreover, there is rising activity noted in the B/Victoria line, which has seen a surge in certain regions, skyrocketing from 6% to over 20% in cases in places such as Hong Kong and the USA.
Sino Biological's Response to Evolving Needs
In response to the WHO's recommendations, Sino Biological has launched a comprehensive portfolio of recombinant antigens aimed at tackling the anticipated flu strains for the upcoming season. This includes the H1N1 HA-trimer from A/Missouri/11/2025, along with its NA and NP proteins, which have shown a purity of over 90% as verified by SEC-MALS, demonstrating the company's commitment to providing high-quality tools for scientific research.
Dr. Rob Burgess, the Chief Business Officer at Sino Biological US, stated, “Our mission is to arm the scientific community with the highest quality tools as swiftly as possible when the emergence of infectious diseases poses a threat to global health.” This statement underscores the urgency with which Sino Biological is approaching vaccine development and its overall commitment to public health.
About Sino Biological
Sino Biological, a globally recognized biotechnology company, specializes in high-quality recombinant proteins, antibodies, and tailored research services. The company supports researchers in over 90 countries, aiding fundamental research, drug discovery, vaccine development, and diagnostics. By leveraging its expansive product portfolio and proprietary quality assurance systems, Sino Biological remains at the forefront of combating infectious diseases and enhancing health outcomes worldwide.
Looking Ahead
While certain statements within Sino Biological's reports may represent forward-looking statements under the Private Securities Litigation Reform Act, the company is clear about the uncertainties and dynamics surrounding vaccine development. As circumstances evolve, so too will their strategies and solutions. Sino Biological is dedicated to keeping stakeholders informed while proactively addressing the public's health needs through innovative scientific advancements.
In conclusion, with the launching of new antigens tailored for the 2026-2027 influenza strains, Sino Biological is not only reinforcing its position as a leader in vaccine development but also reaffirming its commitment to public health and safety during unpredictable flu seasons.
Topics Health)









【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.