
Introducing Innovative SaaS Platform for EPPV Progress Management in Pharmaceuticals
Introduction
Intage Technosphere, a prominent player in the IT sector of marketing research, has announced the launch of an innovative SaaS-based EPPV progress management system. This service aims to support pharmaceutical companies in managing Early Post-marketing Phase Vigilance (EPPV) studies, crucial for ensuring drug safety shortly after market launch.
Background of Development
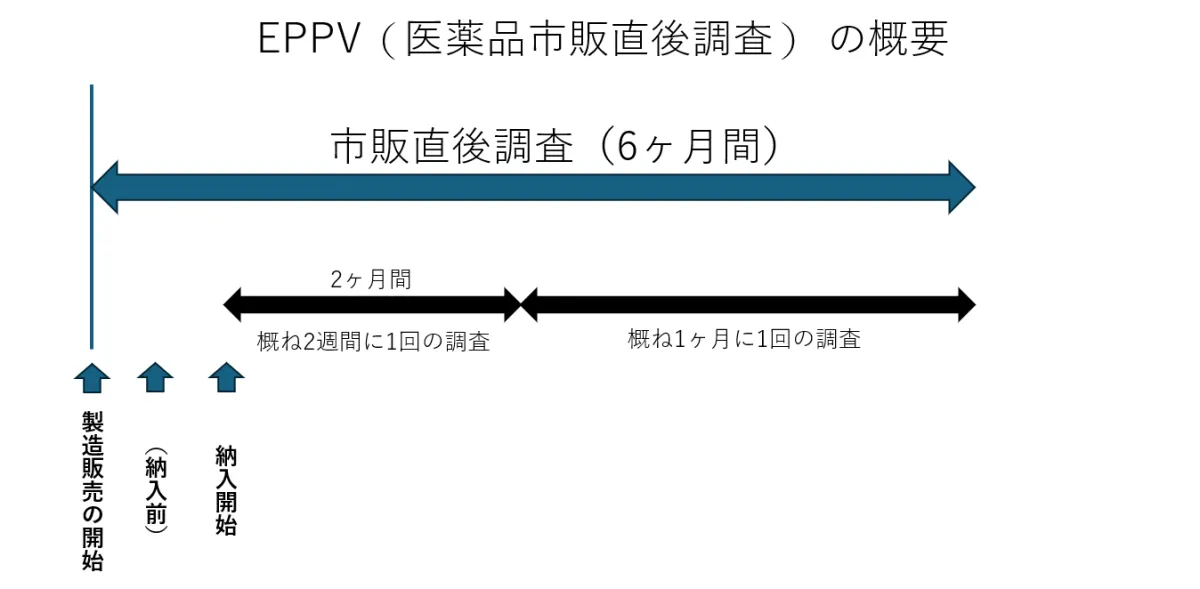
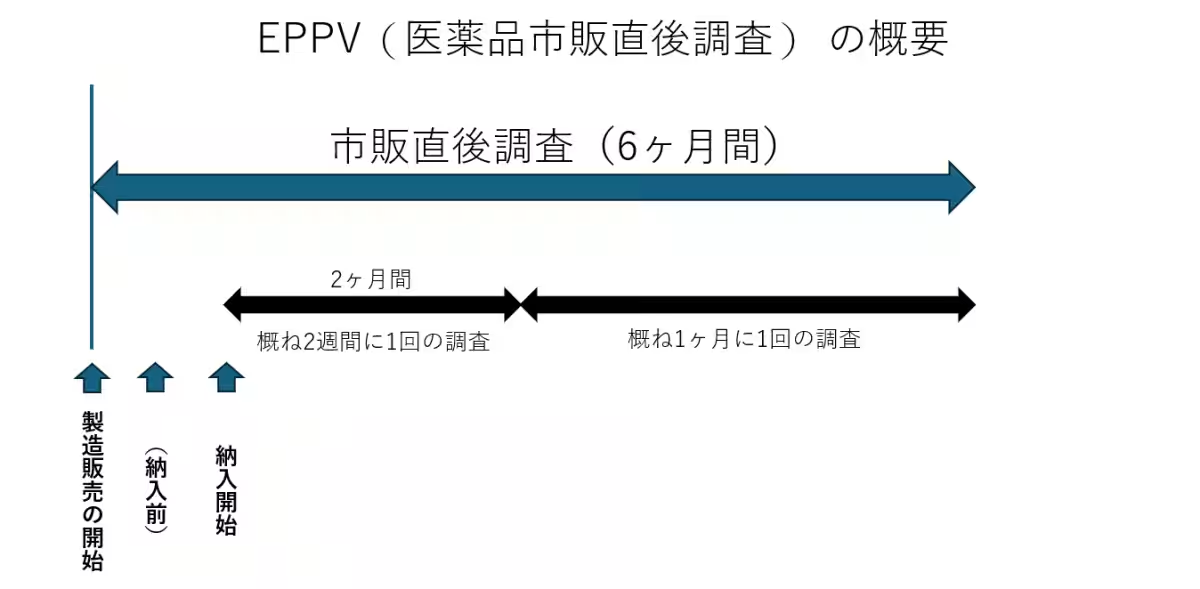
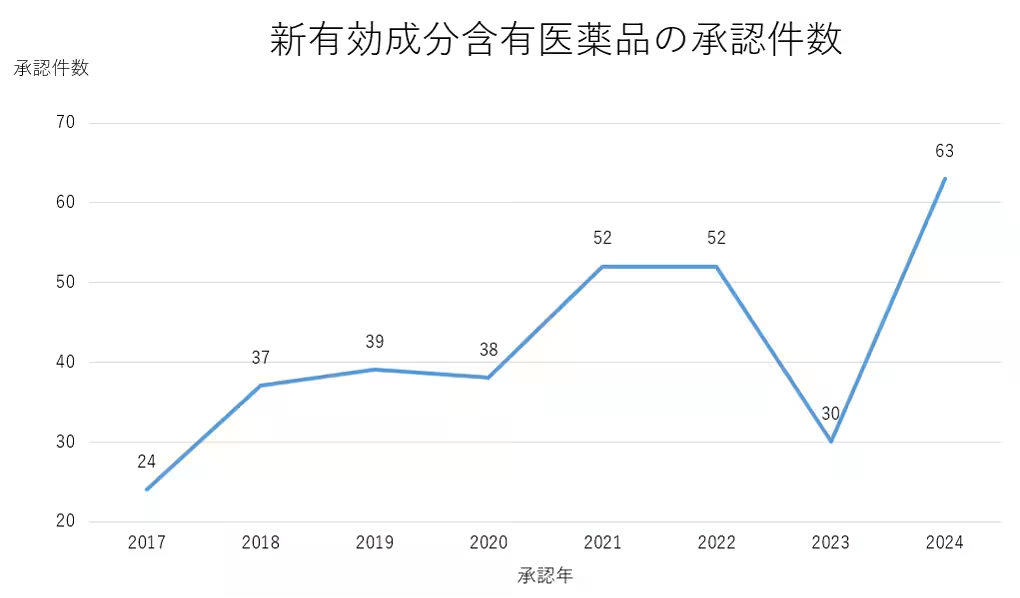
The EPPV is a mandatory safety surveillance process imposed on pharmaceutical companies immediately after a new drug is launched. With the increasing number of new drug approvals featuring innovative active ingredients, the significance of post-marketing safety monitoring has grown tremendously. This SaaS solution responds to the escalating demands of the pharmaceutical sector, allowing for efficient management and oversight without dependence on other systems.
Key Features of the New Service
- - Pre-configured SaaS Model: Unlike previous custom-built services tailored per client, this system is designed to function out-of-the-box with all necessary features.
- - Standalone Usage: EPPV functionalities can be utilized independently, eliminating reliance on daily report systems or other management tools.
- - Flexible Contract Terms: Short-term contracts that align with specific investigation periods are now available.
- - Integrated Progress Management Features: The system comes standard with the essential progress management tools required for EPPV, simplifying data collection and monitoring processes.
Future Developments
Intage Technosphere will showcase this revolutionary EPPV progress management system at the upcoming Pharma IT & Digital Health Expo 2026, scheduled for April 21–23 at Tokyo Big Sight. Attendees will have the opportunity to interact with the platform and view its functionality firsthand. Following this event, the company plans to engage trial users to conduct Proof of Concept (PoC) investigations to validate the system’s effectiveness.
Why This Matters
The pharmaceutical industry is undergoing a rapid transformation, with increasing scrutiny on drug safety and efficacy post-approval. The introduction of this SaaS EPPV progress management platform signifies a crucial step towards enhanced efficiency and compliance within regulatory frameworks.
Intage Technosphere stands firm in its commitment to bridging technology with healthcare needs, fostering advancements in drug safety and monitoring initiatives.
About Intage Technosphere
As a subsidiary of the Intage Group, Intage Technosphere is dedicated to propelling IT initiatives within the marketing research landscape. The company believes in the vision of "Infusing data with soul, inspiring the world through Intelligence, Integrity, and Impression." Among its offerings are AI business applications, software development, database support, and various system operation services designed to improve business processes and assist clients in their data utilization and digital transformation journeys.
For further details on this new service or inquiries, reach out to the public relations team at [email protected] or visit the official website.
As the pharmaceutical industry braces for a future that emphasizes technology in safety surveillance, Intage Technosphere's innovative approach is one that could redefine post-marketing vigilance standards.
Topics Consumer Technology)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.