
Sino Biological Accelerates Influenza Vaccine Development for Northern Hemisphere 2026-2027
Accelerating Vaccine Development: Sino Biological’s Role in Influenza Solutions
In a world that is perpetually challenged by evolving pathogens, the need for timely and effective vaccines has never been more critical. Sino Biological, Inc., a prominent name in recombinant technology, has made headlines by launching a new range of antigens targeting the strains of the influenza vaccine for the Northern Hemisphere slated for the 2026-2027 season. This move comes on the heels of the World Health Organization's (WHO) recent recommendations and underscores the importance of rapid vaccine adaptation in response to ongoing viral mutations.
The WHO has pointed out an alarming shift in the dominant influenza strains, particularly with the emergence of subclade K (J.2.4.1) of the A(H3N2) virus, which was identified in August 2025. This strain has shown increased immune evasion capabilities due to certain mutations, prompting the WHO to designate A/Darwin/1454/2025 as the new reference virus for H3N2. Alongside this, the H1N1 component has been updated to A/Missouri/11/2025, presenting significant implications for vaccine formulation.
Understanding the Current Influenza Landscape
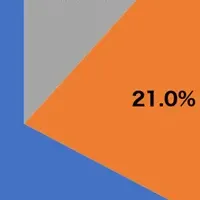
As influenza viruses constantly evolve, maintaining updated vaccines is crucial for effective protection. The newly dominant H3N2 subclade K has transitioned from a minority to a prevailing strain, making it a priority for vaccine development. In addition, the WHO has observed a substantial increase in the activity of the B/Victoria lineage, with proportions skyrocketing from 6% to over 20% in regions like Hong Kong and the United States. In light of these shifts, the WHO has recommended including B/Tokyo/EIS13-175/2025 and B/Pennsylvania/14/2025 in the vaccine composition for the upcoming season.
Sino Biological's Antigen Portfolio
To facilitate global vaccine research and development efforts, Sino Biological has launched an extensive portfolio of recombinant antigens specifically designed for the 2026-2027 influenza vaccine strains. This includes the H1N1 HA trimer, with a verified purity of 90% and above, along with NA and NP proteins. According to Dr. Rob Burgess, the Commercial Director of Sino Biological US, “Our mission is to provide the scientific community with the highest quality tools as swiftly as possible when infectious disease evolution threatens global health preparedness.” This proactive approach exemplifies the company’s commitment to combatting infectious diseases through innovation and support for scientific research.
Sino Biological: A Leader in Biotechnology
As a global biotechnology enterprise, Sino Biological specializes in high-quality recombinant proteins, antibodies, and customized research services. The company serves a vast network of researchers across more than 90 countries, aiding in fundamental research, drug discovery, vaccine development, and diagnostics. Its comprehensive range of products, exclusive quality systems, and innovative research platforms exemplify its role as a key player in the biotechnological landscape.
Conclusion
With the ever-changing landscape of infectious diseases, entities like Sino Biological play a vital role in advancing vaccine technologies. Their swift response to the WHO's recommendations and the launch of specific influenza antigens for the 2026-2027 vaccine underscores a commitment to improving public health and fostering scientific progress in the field of infectious disease control. As we continue to navigate challenges posed by viruses, initiatives such as these will be pivotal in shaping our global health strategies for resilience and preparedness against future pandemics.
Topics Health)









【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.