
Hitachi Collaborates with Shionogi to Utilize Generative AI for Regulatory Document Creation in Pharmaceuticals
Introduction
In a notable collaboration, Hitachi Ltd. has entered into a partnership with Shionogi & Co., Ltd., aimed at revolutionizing the pharmaceutical development process. By leveraging generative AI to assist in the creation of regulatory documents, both companies seek to address challenges faced in the industry, particularly related to the time-consuming and labor-intensive nature of document preparation. As part of this agreement, Hitachi will begin offering this solution to pharmaceutical and healthcare companies in Japan starting in February 2026.
The Solution and Its Impact
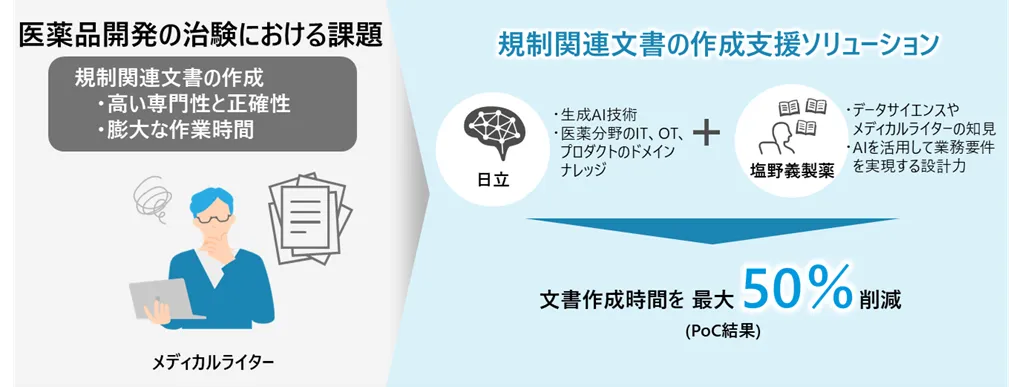
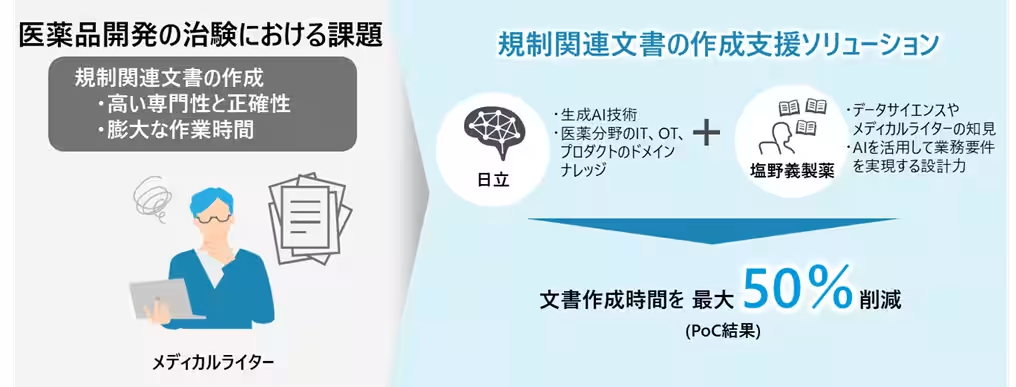
This innovative solution leverages the generative AI capabilities owned by Shionogi, aimed at streamlining the preparation of numerous essential regulatory documents, such as clinical trial protocols and comprehensive trial reports. Traditionally, preparing these documents could take an extensive 3 to 5 months, requiring medical writers to devote between 100 to 280 hours per trial. The solution merges expertise from both firms, employing Hitachi's vast experience in AI deployment and domain knowledge in IT for the pharmaceutical sector, alongside insights from Shionogi’s medical writers and data scientists.
The collaboration has already demonstrated impressive results during its proof of concept (PoC) phase at Shionogi, which confirmed a reduction in document creation times by approximately 50% for comprehensive trial reports and around 20% for clinical trial protocols. Many users noted that they experienced significant overall time savings, highlighting the efficacy of this solution in addressing document complexity and regulatory demands.
Advanced AI Features
The developed solution is designed with user-friendly features, enabling the quick and accurate extraction and summarization of relevant information from a large array of clinical trial data, which often includes a mix of Japanese and English. Additionally, it allows for the rapid creation of initial drafts of regulatory documents. By employing an intuitive interface, this solution can drastically shorten the document preparation process, supporting faster pharmaceutical development while relieving the workload on medical writers.
Future Plans
Looking ahead, Hitachi aims to further enhance this solution under its Lumada brand and explore its applications in the field of biopharmaceuticals. With the increasing complexity of drug development and a dwindling workforce due to demographic shifts, this partnership signifies a crucial step toward enhancing productivity in the pharmaceutical sector.
Hitachi’s Connected Industries (CI) sector is focusing on expanding its next-generation solutions, titled ‘HMAX Industry,’ that integrate advanced AI with rich product installation data. This initiative aims to enhance industry productivity while ensuring a sustainable pipeline of innovative pharmaceutical products. Hitachi's goal is to contribute to better patient outcomes by streamlining drug development timelines and ultimately improving overall healthcare delivery.
Conclusion
In collaboration with Shionogi, Hitachi is at the forefront of revolutionizing drug development through the application of generative AI to reduce documentation burdens. This partnership not only addresses immediate needs in the field but also paves the way for future innovations that could reshape the pharmaceutical landscape, thereby expediting the delivery of new medications to patients and enhancing overall wellbeing.
As the pharmaceutical industry continues to evolve, collaborations like that of Hitachi and Shionogi herald a new era of efficiency and effectiveness, benefitting both companies and the global health ecosystem at large.
Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.