
Cambrex Accelerates Expansion Efforts in the US and Europe with New Manufacturing Facilities
Cambrex Scales Up Production in the US and Europe
Introduction
Cambrex, a global leader in contract development and manufacturing (CDMO), has made significant strides in its expansion efforts in both the United States and Europe, recently announcing the completion of initial engineering studies for a new active pharmaceutical ingredient (API) manufacturing facility in Charles City, Iowa. This monumental step marks progress towards the company's previously announced $120 million investment aimed at enhancing API production capacity in the United States. The groundbreaking ceremony for the new manufacturing plant is scheduled for the end of 2026.
U.S. Expansion: A New Chapter in Iowa
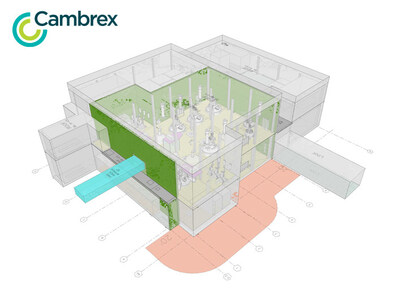
The anticipated expansion in Charles City involves a state-of-the-art facility capable of accommodating 140,000 liters, equipped with both large- and medium-scale reactors, cutting-edge Hastelloy filter dryers, and improvements to existing production facilities. Once operational, this expansion is expected to boost the site's large-scale manufacturing capacity by 20%. The facility is designed to support complex chemical processes, including controlled substances, highly potent active pharmaceutical ingredients (HPAPI), and commercial-scale liquid peptide production, reinforcing Cambrex's status as a trusted partner for innovative pharmaceutical companies.
Investments Across the Atlantic: The Italian Initiative
Not resting on its laurels, Cambrex is also making waves across the Atlantic with a $30 million expansion at its plant in Milan, Italy. This investment will incorporate new analytical development and process R&D capabilities along with upgrades to multiple production facilities. The R&D expansion is slated for completion in the second half of 2027, and the company has already acquired additional land to support future growth and increasing demand for CDMO services.
Words from Leadership
Claudio Russolo, Chief Operations Officer, expressed, "Our Milan facility has a storied history of supporting pharmaceutical product development and manufacturing, celebrating its 80th anniversary this year. We are fully committed to ongoing investment to meet the growing market demands for complex chemical compounds, including high-potency molecules and controlled substances."
Commitment to Excellence
Cambrex's continued investment is a testament to its dedication to providing advanced solutions for the development and manufacturing of complex small molecules. With a scientific approach underpinning its operations, Cambrex is firmly positioned to remain at the forefront in supporting the next generation of transformative therapies. The company’s sophisticated knowledge base, combined with operational excellence, ensures its leading role in this evolving landscape.
About Cambrex
Cambrex is a premier global contract development and manufacturing organization (CDMO) that provides comprehensive pharmaceutical development and manufacturing services throughout the drug lifecycle, along with integrated analytical services and support for Investigational New Drug (IND) applications. With 45 years of experience and a continuously growing team of over 2,000 experts serving clients globally across North America and Europe, Cambrex offers a wide range of specialized technologies and capabilities in active pharmaceutical ingredients, including continuous flow, controlled substances, peptide synthesis, solid-state science, material characterization, and HPAPI.

Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.