
Breakthrough BNCT Treatment Initiated in China's Hainan Medical Zone
Introduction
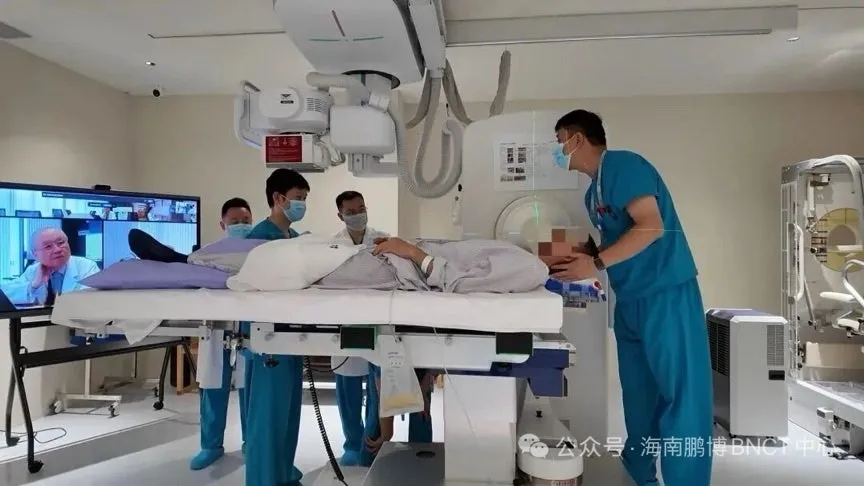
On March 19, 2026, a significant milestone was achieved in cancer treatment with the commencement of Boron Neutron Capture Therapy (BNCT) targeting head and neck cancers at the Pengbo (Hainan) BNCT Center located in Boao Lecheng International Medical Tourism Pilot Zone, Hainan, China. This pioneering treatment utilizes the BNCT system "NeuCure®" developed by Sumitomo Heavy Industries and is supported by the BNCT dose calculation program "NeuCure Dose Engine®". The fast-tracked clinical application of this technology represents a shift in cancer treatment protocols in China.
Overview of BNCT Treatment
The BNCT approach integrates the "NeuCure®" system with the boron pharmaceutical "Steboronin®" developed by Stellar Pharma Inc., marking the first implementation of BNCT treatment in real clinical settings in China. The collaboration signifies not only a technological advancement but also a strong commitment by Sumitomo Heavy Industries to broaden its market reach in China. This initial treatment is viewed as a launchpad for further developments and expansions within the healthcare sector.
Clinical Data and Future Prospects
Importantly, the clinical data gathered from the Hainan Medical Zone will likely play a pivotal role in future applications for medical device sales approvals across mainland China. Recognizing the potential of this therapy in improving patient outcomes, Sumitomo Heavy Industries is strategically positioning this treatment as the first step in penetrating the Chinese market. As part of its strategy, the company aims to collaborate with various partners, thereby ensuring that advanced cancer treatment technologies are disseminated internationally, contributing to the progression of global healthcare.
Hainan Medical Zone
The Hainan Medical Zone is designed as a special regulatory area focused on promoting medical tourism through relaxed regulations. In this unique environment, preferential policies for the import of medical devices and pharmaceuticals are in place. One of the most notable features of this initiative is the allowance for importing and using medical devices and pharmaceuticals that have been approved in other countries, even in cases where they are not yet authorized domestically in China. This flexible regulatory approach allows for urgent medical needs to be met without the typical burdens of undergoing extensive clinical trials. Healthcare institutions specified for such uses can import these essential treatments, ensuring timely access for patients.
Conclusion
The recent commencement of BNCT treatment in Hainan reflects a significant advancement in cancer care and highlights the dynamic evolution of healthcare regulations in China. The integration of innovative treatment technologies like "NeuCure®" and the strategic collaboration with pharmaceutical companies represent an exciting phase for both domestic and international markets. The hope is that the successful application of BNCT will pave the way for similar advancements in other areas of healthcare, making a real difference in the lives of patients facing the challenges of cancer.
For more details, refer to the official announcement regarding the introduction of BNCT in Hainan: Link to Announcement

Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.