
Innovative Nanotube Membrane Stamp Technology Enhances Cell Functionality by 25%
Introduction
Researchers at Waseda University have developed a cutting-edge technology utilizing a nanotube membrane stamp that allows for the direct transfer of cellular liquids and mitochondria between cells. This technique, touted as a new "cell surgery" method, has demonstrated the capacity to enhance cell functionality by up to 25%, creating substantial implications for fields like cell therapy and regenerative medicine.
Key Developments
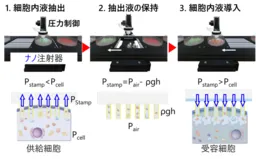
The collaborative study led by Professor Takeo Miyake has achieved a remarkable transfer efficiency of approximately 90%, with a cell survival rate around 95%. The technology's advanced pressure control mechanism in the nanotube syringe enables researchers to efficiently extract and reintroduce vital cellular components while maintaining their viability.
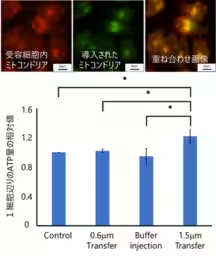
The ability to transfer mitochondria—critically important cellular organelles responsible for ATP production—has been quantified for the first time. This innovative procedure not only facilitates the transfer of such organelles but does so in a manner that notably increases ATP production within recipient cells.
Background of Research
Cellular communication is primarily executed through the movement of proteins, RNA, and organelles. The phenomenon of mitochondrial transfer via tunneling nanotubes (TNTs) has been observed in nature, influencing energy metabolism and disease progression. Historically, manually controlling these transfer processes has been challenging due to low occurrence rates and dependence on specific cell types or conditions.
Existing cell manipulation technologies have largely focused on directing materials into a cell rather than facilitating two-way transfer between cells. Techniques like viral vectors, electroporation, and liposome-mediated delivery have served well for introducing components into cells but have not been able to extract and transfer components efficiently and reliably across multiple cell types.
Research Outcomes
The newly developed nanotube syringe system represents a shift from random natural occurrences of cell content transfer to a repeatable and quantitative process. By integrating controls for extraction, holding, and introduction into one platform, the researchers have created a reliable method for intercellular transfer. The nanotube structure, made from gold and glass, adeptly manages the flow of cellular components either by lowering pressure to draw liquids into the nanotube or by applying positive pressure to transfer them into target cells.
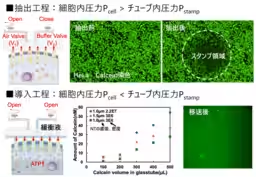
The study confirmed a high efficiency of cytoplasmic transfer when using HeLa and NIH-3T3 cells across homogenous and heterogeneous pairings, maintaining both a high transfer efficiency and cell viability.
These findings indicate that this technology can facilitate the exchange of essential cellular components without detrimental effects on cellular functions.
Enhancements in Cellular Functionality
The researchers further examined the nanotube's diameter to optimize mitochondrial transfer. While narrower nanotubes showed limited transfer capabilities, a diameter of 1.5 μm proved ideal, allowing effective extraction of mitochondria and subsequent transfer to recipient cells. The result was a significant uptick in ATP production within just 24 hours post-transfer, showcasing the technology's efficacy in enhancing cellular functioning.
Societal Impact
The solidification of reliable, reproducible procedures for intercellular component transfer opens avenues for significant advancements in cellular research and therapy. By standardizing methodologies within this field, it is anticipated that the quality and reliability of cell manipulation techniques will experience notable improvements.
Future Directions
The potential applications of this nanotube injection technology in regenerative medicine remains expansive. Future research will focus on testing various cell types for reproducibility and stability, with a vision for collaboration with pharmaceutical and research entities to explore applications beyond animal cells, including those from plants and microorganisms.
Conclusion
This pioneering nanotube membrane stamp technology stands at the frontier of cellular research and manipulation, promising to reshape the landscape of cell therapy and regenerative medicine through increased efficiency and functionality in cellular component transfer.

Topics Health)










【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.