
Understanding Mitochondrial Changes Induced by Tau Protein in Dementia to Break the Cycle of Neuronal Death
Understanding Mitochondrial Changes Induced by Tau Protein in Dementia
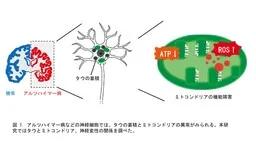
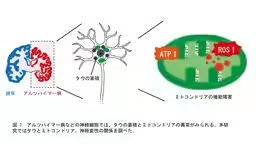
Neurodegenerative diseases, including Alzheimer's disease, are characterized by the accumulation of abnormal tau protein in the brain, leading to progressive neuronal loss. While it is known that tau contributes to neuronal death, the underlying mechanisms are still not thoroughly understood. In recent research conducted by graduate students Arisa Tamura and Marie Noguchi from Tokyo Metropolitan University, along with Professor Kanae Ando, it was discovered that tau disrupts mitochondria's functioning, instigating a cycle of neuronal death.
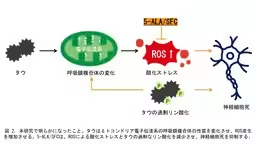
Using a Drosophila model that expresses human tau, the researchers observed a decrease in the brain's ATP levels coupled with heightened oxidative phosphorylation (OXPHOS) activity, resulting in increased production of reactive oxygen species (ROS). The introduction of a combination of 5-Aminolevulinic Acid (5-ALA) and Sodium Ferric Citrate (SFC) showed promise in mitigating oxidative stress, reducing excessive tau phosphorylation, and alleviating tau-induced neurodegeneration. This study suggests that the 5-ALA/SFC combination could potentially interrupt the negative feedback loop between tau, mitochondrial dysfunction, and oxidative stress, thereby suppressing neurodegeneration.
Background of the Study
Mitochondria, known as the powerhouse of cells, are essential for normal neuronal function. They produce adenosine triphosphate (ATP) via the electron transport chain. However, this process can lead to the generation of harmful ROS, particularly when mitochondrial proteins involved in OXPHOS are dysfunctional. Mitochondrial impairment has been reported in neurodegenerative diseases, including Alzheimer's, pointing towards its significant role in disease pathogenesis.
In the brains affected by these diseases, tau protein accumulates abnormally. Tau is a microtubule-associated protein which, under normal circumstances, does not adopt any specific structure. However, in pathological states, it becomes hyperphosphorylated, aggregates, and accumulates in neurons, leading to cell death. Understanding how tau inflicts damage on neurons and the implications for mitochondrial function has been a primary focus of this research.
Detailed Findings of the Study
The researchers assessed mitochondrial changes by expressing human tau in Drosophila's neuronal and photoreceptor cells. Measurement of ATP levels revealed a decline due to tau expression, while the total amount of mitochondria did not decrease. Interestingly, the expression of proteins involved in the respiratory chain was upregulated, and the activities of complexes I and IV were enhanced. However, there was a marked decrease in the distribution of mitochondria to neuronal processes in Drosophila expressing tau.
Additionally, using a reporter system that specifically detects mitochondrial ROS, they found an increase in ROS levels within the neurons. Experiments where tau was expressed in photoreceptor cells demonstrated that neuronal death occurred, although feeding these flies a diet enhanced with 5-ALA/SFC mitigated this effect. Notably, the administration of 5-ALA/SFC also decreased tau phosphorylation at disease-relevant sites, suggesting a protective effect.
Even though the treatment didn't significantly improve overall mitochondrial function or distribution, it did substantially reduce oxidative stress, highlighting its potential as a therapeutic intervention.
Significance and Implications of the Research
The risk of developing dementia-related neurodegenerative diseases increases with age, and with an aging population, the need for effective preventive and therapeutic strategies is crucial. This research sheds light on the mechanisms behind the pathology of such diseases. Particularly noteworthy is the discovery that tau alters the activity of the respiratory chain and increases ROS production.
Oxidative stress, which damages proteins, lipids, and nucleic acids can instigate various stress responses within cells. Some enzymes activated by oxidative stress are known to provoke tau phosphorylation. The findings from this study indicate that despite the absence of significant recovery in mitochondrial function, the reduction of oxidative stress, decreased tau phosphorylation, and the protection against neuronal death occur due to the administration of 5-ALA/SFC.
This suggests a vicious cycle where changes induced by tau in the respiratory chain increase ROS production, which further exacerbates tau phosphorylation, leading to cellular damage. The study proposes that 5-ALA/SFC can disrupt this cycle by diminishing oxidative stress, thus contributing to the attenuation of neurodegeneration. Such insights are instrumental for developing preventive and therapeutic measures against tau-related neurodegenerative diseases.
Conclusion
The research conducted provides a deeper understanding of how tau protein impacts mitochondrial function and neuronal health, unveiling potential protective strategies through anti-oxidative therapies. As the search for effective treatments for dementia progresses, findings from this study promise to influence future research directions and therapeutic developments.
References
1. Tamura, A., Noguchi, M., Nozawa, N., Suzuki, E., Ando, K. (2026). 5-ALA/SFC Mitigates Tau Toxicity via Lowering Oxidative Stress in a Drosophila Model of Tau Toxicity. Life.
DOI: 10.3390/life16050725
Link to the study.
Topics Health)









【About Using Articles】
You can freely use the title and article content by linking to the page where the article is posted.
※ Images cannot be used.
【About Links】
Links are free to use.